Late addition (July 2019)
Please forgive this postscript, correction, “prescript”, correction, intrusion, added many years later – based on some 350 and more postings here and elsewhere.
That’s including some 7 years of my hands-on investigation into image-forming techniques, chosen to be credible with simple, indeed crude, medieval (14th century) technology etc etc.
(Oh, and yes, I accept the radiocarbon dating, despite it being restricted to a single non-random corner sample, making all the oh-so-dismissive, oh-so-derogatory statistics-based sniping totally irrelevant – a ranging shot being just that me dears- a single ranging shot, albeit subdivided into three for Arizona, Oxford and Zurich).
Sindonology (i.e. the “science” , read pseudoscience – of the so-called “Shroud ” of Turin) can be simply summed up. It’s a re-branding exercise, one designed to pretend that the prized Turin possession is not just J of A’s “fine linen”, described in the biblical account as used to transport a crucified body from cross to tomb.
Oh no, it goes further, much further, way way beyond the biblical account. How? By making out that it was the SAME linen as that described in the Gospel of John, deployed as final “burial clothes”. Thus the description “Shroud” for the Turin Linen, usually with the addition “burial shroud”. Why the elision of two different linens, deployed for entirely different purposes (transport first, then final interment)?
Go figure! Key words to consider are: authentic relic v manufactured medieval icon; mystique, peaceful death-repose, unlimited opportunity for proposing new and ever more improbable image-formation mechanisms etc. How much easier it is to attach the label “Holy” to Shroud if seen as final burial clothes, in final at-peace repose – prior to Resurrection- as distinct from a means of temporary swaying side-to-side transport in an improvised makeshift stretcher !
As I say, a rebranding exercise (transport to final burial shroud) and a very smart and subtle one at that . Not for nothing did that angry local Bishop of Troyes suddenly refer to a “sleight of hand” after allegedly accepting it when first displayed. Seems the script was altered, or as some might say, tampered with! It might also explain why there were two Lirey badges, not just one. Entire books could be written on which of the two came first… I think I know which, with its allusion (?) to the Veil of Veronica… yes, there are alternative views (the face above “SUAIRE” a visual link to the face-only display of the Linen as the “Image of Edessa” or as that on the then current “Shroud” per se.

Face shown (left) on mid- 14th century Machy Mould (recently discovered variant of the Lirey Pilgrim Badge) above the word “SUAIRE” (allegedly meaning “shroud”). Inset image on the right: one version among many of the fabled “Veil of Veronica” image. I say the two are related, and deliberately so, but this is not the time or place to go into detail.
No, NOT a resurrectional selfie, but instead a full size version of, wait for it, the legendary VEIL OF VERONICA , product of inital body contact – no air gaps- between body and fabric, but with one important difference. The Turin image was intended to look more realistic, less artistic.
How? By displaying a negative tone-reversed image implying IMPRINT (unless, that is, you’re a modern day sindonologist, in which case ‘resurrectional proto-photographic selfie” becomes the preferred, nay, vigorously proferred explanation assisted by unrestrained imagination, creation of endless pseudoscience etc etc, with resort to laser beams, corona discharges, nuclear physics, elementary particles, earthquakes etc etc – the list is seemingly endless!
Welcome to modern day sindonology.
Personally, I prefer no-nonsense feet-on-the-ground hypothesis-testing science, aided by lashings of, wait for it, plain down-to-earth common sense.
Start of original posting:
The original introduction here, setting out my strategy for dealing for Ray Rogers’ formidable set of FAQs has now been deleted as being ‘past their sell-by-date’. It is now 16:40, Wednesday 9th Jan, all the major points of contention (and occasional agreement) have been dealt with.
Right now my time would be better spent in following up the new discovery made with the microscope, namely the (apparent) preferential scorching of the nodes in linen fibres and its implications – see current post that immediately follows this one.
As far as the FAQs are concerned, I hope to return in a day or two, and create a new re-organized posting in which the FAQs are dealt with in the same order as in Rogers’ FAQs. My posting below has them the other way round which some may find irritating, a result of the decision to stagger the work, rather present a huge slug of script as a fait accompli , which means they appeared in reverse chronological order.
The new posting will have a few afterthoughts, cosmetic tidying up etc that are not in what appears here. It will also have a snappier title, the present one criticized else as ‘cumbersome’. Apologies then for deserting the present task prematurely, but rest assured I have not abandoned the principles that I learned as a child recalling the frontispiece of a charming 1950s book I received from the US side of the family, crammed with inventive home projects for the easily bored child. It read:
“If a task is once begun
Never leave it till it’s done
Be the labor great or small
Do it well or not at all”
Now to Ray Rogers and his FAQs.

A pastiche of relevant screen grabs to set the scene…
In time, I hope to address all 19 points of Raymond N.Rogers much-cited FAQs (Frequently Asked. Questions), while making no secret of wishing them to become FQAs (Frequently Questioned Answers).
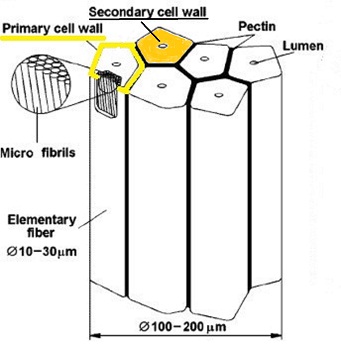
Schematic diagram: bundle of bast fibres in flax stem. Spot the easy-to-overlook primary cell wall, highlighted in yellow. It is these bast fibres which, after retting to microbiologically digest the cementing pectin matrix,become the uncemented fibres comprising linen yarn, with primary cell walls still present, but scarcely visible due to the much thicker secondary cell wall (orange) – the latter predominantly highly ordered cellulose microfibrils.
(End of introduction.)
A detailed critique now follows. I have chosen Rogers’ own FAQs as source material, at least for starters, being reasonably concise, and accessible without charge as a pdf document. The chief problem is knowing which of the 19 FAQs to include and exclude, and in which order to tackle them. I have opted to deal with most if not all in the same order as Rogers, even though that means dealing with issues that are not immediately relevant to the main issue addressed here, namely Rogers’ proposed mechanism by which the Shroud acquired its superficial image involving a hypothesised “impurity layer”, without (as we shall see) so much as a nod in the direction of the primary cell wall. By taking them in the same order, I avoid the charge of being selective in my choice of material, or of deviating from Rogers’ order of presentation. If crucial details appear to be omitted from crucial arguments, that will be because Rogers chose to withhold them, not me, and those that he considered important simply appear later rather than sooner. I shall have to make frequent use of the “read on” tag to keep the posting to a reasonable length.
The image is extremely faint and difficult to see. Much more detail can be seen in contrast-enhanced and ultraviolet photographs; however, they are somewhat misleading to studies on image formation.
Yes, but as noted earlier, that did not prevent Rogers from choosing a uv-picture to demonstrate the banding or quilting effect, upon which he then placed so much reliance in making a pitch for the image NOT being on the linen itself (cue his “impurity layer”)
Whatever produced the image color did not produce very much color.
Probably a lot more was visible centuries ago when it was on public display in the open air, town squares etc with throngs of thousands etc. One hardly imagines that folk would have travelled from far and wide to see something that was scarcely visible unless at a carefully measured five feet. So ideas about image formation must not get too hung up on the idea of extreme superficiality, or the claim that conventional science is incapable of explaining its present superficiality. Stuff happens. Images fade. Conventional science does not have access to a time machine , or a bottle of “New-Formula Magic Restore”, guaranteed to reverse the effects of centuries of exposure to light, oxygen, fires, pollutants etc etc. (“Repeat application if necessary”)
Scientific observations were planned in order to learn as much as possible about the image distribution and chemical composition.
Really? So how come we know next to nothing about its distribution (on top of or under the bloodstains –so far investigated by one approach only, a wet method using proteolytic enzyme) or its chemical composition (pyrolyis product? Maillard product? something else?)
The Shroud was observed by microscopy, visible and ultraviolet spectrometry, infrared spectrometry, x-ray fluorescence spectrometry, and thermography.
So how come I have never seen a decent photomicrograph of the Shroud in visible light- say x 40 magnification, at the level of individual fibres (note: fibres, not threads), never mind uv pictures etc.
Later observations were made by microchemistry, petrographic microscopy, scanning-electron microscopy, energy dispersive x-ray analysis, pyrolysis-mass-spectrometry, and laser-microprobe Raman analyses.
Hand up all those of you who have seen a scanning electron micrograph of the Shroud, especially those bloodstains, correction, “serum exudates from retracted blood clots” etc etc..?
But in other respects a good range of techniques. Who was asked to apply them? Los Alamos colleagues presumably. What contribution if any were they allowed to make to the final STURP conclusions? Or were they merely used as backroom boys at the Los Alamos research institute “on tap”, harnessed for their technical know how? I bet they loved that – scientists being used as technicians.
Without making any assumptions that are based on the appearance of the image, some statements of fact can be proved from the scientific observations.
One establishes a fact in science beyond all reasonable doubt, ie. making it generally or universally credible. Proof on the other hand is something that should be left to the mathematicians, who are able to load the die by taking ‘self-evident’ axioms as their starting point . Scientists are usually loath to take anything as self-evident. As a generalisation, all generalisations are dangerous, including this one…
1) Reflectance spectra, chemical tests, laser-microprobe Raman spectra, pyrolysis mass spectrometry, and x-ray fluorescence all show that the image is not painted with any of the expected, historically-documented pigments and media.
“Expected”? “Historically documented”? Was a lawyer called in to help draft that statement (not that I personally disagree with it, but others might)
2) No painting pigments or media scorched in image areas or were rendered water soluble at the time of the AD 1532 fire.
There seems to be a typo there – making it difficult to extract the precise meaning.
3) Direct microscopy showed that the image color resides only on the topmost fibers at the highest parts of the weave.
So how come shortwave electromagnetic radiation – uv, corona discharges etc- is still being touted as the source of energy, acting across across sizeable air gaps (the oft cited/invoked “cloth-body distance” )? What difference would it make if one thread were slightly higher in the weave than others, while still in the ‘line of fire’ so to speak? To Rogers credit he dismissed radiation. ( So did I, after briefly entertaining the idea of scorching by radiation – from radiated heat – provided an absorbing pigment was present, e.g. charcoal to act as a thermosensitizer that could later be washed out. That idea was quickly discarded when it was pointed that the ‘charcoal artist’ would have had to draw a light/dark reversed negative – which was hardly likely).
4) The color density of any specific image area depends on the batch of yarn that was used in its weave. The cloth shows bands of slightly different colors of yarn.
OK as far as it goes. But elsewhere it goes too far.
5) Adhesive-tape samples show that the image is a result of concentrations of yellow/brown fibers.
Caveat: yellow/brown has to be read as a single hue, between yellow and brown, not as either yellow or brown. Otherwise it contradicts the claimed ‘half-tone’ effect which says that image density is due to differences in the number of single-hue fibres per unit area, not to differences of hue between fibres.
6) The image does not fluoresce under ultraviolet illumination.
Caveat: it may have done originally. Some fluophores/fluorochomes from,say, pyrolysis reactions ( furfural etc) are liquids at room temperature with small but appreciable vapour pressures that would gradually evaporate over long periods of time and/or become oxidized etc with loss of fluorescence.
7) The image of the dorsal side of the body shows the same color density and distribution as the ventral, and it does not penetrate the cloth any more deeply than the image of the ventral side of the body.
Worth keeping in mind when assessing different theories of image formation (and hardly favourable to Rogers’ own preferred ‘diffusion’ theory if it needs thermal convection as well to move ‘orthogonally’ and/or prevent excessive spread. Thermal convection is always upwards, so would surely produce a frontal image that was darker).
More to come. Logging off at 14:40 Tuesday
(Oops, I inadvertently skipped a section. Quite how I’m not sure, but Rogers’ own reorganization, rolling of two FAQs into one, may have been a factor.
Anyway, today (Wed 9th Jan) I shall proceed to address the missing part, which as it happens dovetails neatly with my current research interest, so will more fun to write).
Flax fibers look like small lengths of bamboo under a microscope.
Yes, this blogger is very interested in the microstructure of flax and linen fibres right now, especially the regions that give the bamboo-like appearance, i.e the nodes, aka dislocations. Might they be the reported sites of mechanical weakness in TS image fibres, and if so, why?
The gross internal composition of a flax fiber is shown in the figure (after Cardamone).
The pdf document shows this illustration. “Cardamone” is apparently a plant anatomist. Her first name escapes me, but will be added later.

Screen grab from Rogers’ FAQs, no. 16, with Cardamone’s diagram of a cemeneted bundle in situ bast fibres in a flax stem from which linen fibres derive after lengthy processing (retting etc). Note: this diagram is not to be confused with a so-called ‘elementary’ flax or linen fibre.
That’s a curious and potentially misleading choice of diagram. It certainly gave this blogger a lot of difficulty when trying to get his head round the difference between flax fibre and linen, i.e. before and after retting. That is a botanist’s diagram, before retting. It shows bundles of elementary fibres held together with a pectin cement. It might be called a flax fibre, or better still bundles of in situ bast (i.e. stem) fibres but should not be confused with the elementary fibres of linen, of which there may be a 100 or more per thread, uncemented needless to say. At the scale of that diagram, there are no nodes labelled, nor cell walls. So despite showing all those dividing ‘partitions’ (my term) it’s impossible to distinguish between nodes, of which there are hundreds per (elongated cell) or the infrequent walls that separate one cell from another.
Here’s a photograph I took yesterday of nodes in linen. Scorching shows up the nodes better (an unexpected and intriguing finding that I shall be following up).

Those darker cross-sbands (nodes, aka dislocations)in this lightly scorched specimen may look like partitions or barriers dividing up the elongated fibre cells, but there is in fact a continuous fibre running though all of them, in the form of the secondary cell wall, comprising many cellulose microfibrils. The internal structure of flax and linen fibres is more complex than it may seem at first glance, and there’s probably a lot we don’t know even now, especially regarding the structure and function of those mysterious nodes, often referred to as growth nodes, albeit it with little or no justification.
As I say, all very confusing at least to novices, which is what this blogger was on the subject until a week or two ago. It’s an uncharitable thing to say I know, but I strongly suspect that Ray Rogers never got beyond the novice stage when it came to flax and linen microstructure. If anyone knows otherwise, and wishes to correct the record, then please get in touch.
The cellulose molecules in flax fibers are folded back and forth in a fairly regular arrangement, and they show the properties of crystallinity. The fibers are composed of closely packed “ultimate cells” of the fibrillar structure that are cemented together with holocellulose and lignin. You can see the ultimate cells under a microscope, and abraded fibers often show ultimate cells sticking away from the surface. These were the structures that were mistaken for “filamentous bacteria” by Garza- Valdes.
This section can be highly confusing to a newcomer on account of the way it jumps back and forth between different levels of organization, introducing a (rarely used?) term “ultimate cell” which is undefined. Thus it begins at the molecular level in the first sentence, it then jumps to fibres which are macroscopic, and then down a level to those ‘ultimate cells’ (with no reference to their shape or size), it then goes to “fibrillar structure” (undefined) which is a lower level than “ultimate cells”, then back to fibres, and then a tangential reference , apparently, to the ‘bioplastic’ contoversy with that mention of ‘filamentous bacteria’
As for that mention of “holocellulose”, words fail me. That’s a chemist’s term, mainly used in the timber industry for wood that has been stripped of lignin and other odds and sods (“extractives”) to leave a mixture of polysaccharides that might be described as cellulose+, the plus being any hemicelluloses, pectins etc that were still around. “Holocellulose” in this context means cellulose plus non-cellulosic polysaccharides, those two being chalk and cheese in terms of physical and chemical properties. So what’s that quaint and opaque term “holocellulose” doing in a set of so-called FAQs intended for the general reader? I shouldn’t say this, I know, of someone no longer here to defend himself, but oh my, it is quite difficult at times to escape the impression that Mr. Rogers enjoyed all the attention of being STURP’s star turn (“world-renowned chemist etc). “Holocellulose” is just a tiny symptom of what I see as occasional grandstanding, slipping in ‘teccie’ terms that would require the diligent reader to nip off and consult a dictionary, or, as I have just done, to a search engine, needing to refresh my memory as to the precise meaning of “holocellulose”. Most of his FAQ-readers would probably have moved quickly on, thinking “Cor, this guy really knows his stuff”. But that’s the trouble. There are/were occasions when there’s more than a hint that Ray Rogers did not have the grasp of every relevant detail that he would have had us believe, especially outside of the kind of chemistry he did for a living (testing the safety of chemical explosives etc).
It has taken this blogger hours to resolve this hotch-potch of detail, so much of it confusing, sufficient to picture in his mind a simplified hierarchy of organization.
When you rotate the stage of a petrographic microscope with crossed polarizers while looking at a flax fiber, straight lengths change from black to colored every 45?. The fiber is birefringent and has an ordered structure. Most of the cellulose of the fibers is in a crystalline structure. In structures like flax, it is called a “fibrillar” structure.
Yes, though it might have been more novice-friendly to point out that fibrils are linear assemblies of highly organized cellulose that comprise the thick secondary cell of flax and linen fibres, accounting for most of the diameter of the elementary fibres which in bundles we call the thread. These fibrils, which are subcellular entitities, and disposed as helices, run the entire length of the elongated cells of elementary fibres, which can be several centimetres in length, the latter (individual fibre cells) being fused end-to-end. Admittedly I have yet to see a diagram that conveys all this information in a form intended for us Shroudies, as distinct from botanists.
McCrone ignored our agreements for work on the STURP sampling tapes: he stuck them all down to microscope slides. This made observations much harder; however, flax and cotton fibers can still be distinguished by their indexes of refraction.
OK, so that was naughty of Walter McCrone, but what’s a side swipe doing in FAQs against a fellow scientist. (Rebuttals of FAQs are different matter altogether where scientific etiquette is concerned ;-).
Returning to the teccie stuff: flax and cotton fibres should be easily distinguishable without resorting to refraction, since they have a distinctive, i.e. twisted ribbon-like appearance, and being seed hairs as distinct from stem support fibres, are said to be entirely single cells.
Crystallographic observations must be made on the specific fibers that reach extinction at the same angle as the tape (while everything is black). The index of refraction of a normal linen fiber parallel to its length is nearly identical to that of the adhesive on the sampling tapes (it nearly disappears). That index is very close to 1.515. The index across the fiber is appreciably lower than the adhesive. The indexes of refraction and crystallinity of image fibers are identical to unaffected fibers. Bent, crushed, or otherwise damaged fibers show strain dichroism and will give an erroneous index. Most flax fibers show intense birefringence colors when they are viewed at a 45º angle from the plane of polarization of the microscope.
Cotton has a low birefringence, usually appearing white (first-order white), and it is a thin, wide tape that shows periodic reversals (twists).
Again, at the risk of sounding monotonous, FAQ writers really ought to work on a ‘need-to-know’ basis’ (or have a Editor with a scary face).
Response will come later (presently 17:10 Sunday)
Yawn: this task is becoming seriously depressing – watching an otherwise competent chemist dig himself into an ever deeper hole – the result of tunnel, nay borehole vision (vertically downwards). There are those who have survived him who exhibit the same kind of single-mindedness to the point of obsession, persisting as they do without objective evidence that they can see coins, flowers or lettering in photographs of the Shroud image.
There’s a splendid paper by Paolo Di Lazzaro, Daniele Murra and Barrie Schwortz which Dan Porter has just flagged up on his site, exploring the phenomenon in depth of the way in which the brain can play tricks- taking inappropriate clues from the surroundings that may cause one to “see things that are not really there” (pareidolia)
It shows the so-called ‘checker optical illusion’. You are advised to view the checker board through cut-out paper masks to confirm that your eyes are deceiving you into thinking square A is dark grey and square B off-white (the latter is dark grey too!). The mask works, but is a bit fiddly and unsatisfying. There’s a more striking way of demonstrating the effect to yourself and company at the same time – simply put the checker board into Microsoft Paint, as I have just done, and then use the eraser to blank out the surrounding squares one at a time. Watch Square B gradually have its “real” darkeness restored, finally to become the same as square A.
Amazing!
……………………………………………………………………..
Update: Monday 7th Jan: back to the task in hand. Time now to address FAQ 15 (must endeavour to keep my comments brief in view of the numerous points raised, especially as they are not germane to the central issue – i.e. that alleged “impurity layer”, and which are addressed here purely for the sake of completeness – and to convince the reader there is nothing I have overlooked or avoided. There are some uncharitable folk who seem to have got it into their heads that I am selective with my choice of “targets” – deliberately ignoring important pro-authenticity data or claims – to which my reaction is one of utter contempt).
13:00 Monday 7th Jan Response in preparation/instalments now follows.
Although the fire of 1532 nearly destroyed the Shroud, it created opportunities for many types of chemical studies.
Nearly destroyed? That’s assuming that the fire was accidental, that the temperature really did get high enough to melt the silver reliquary. There’s another possibility – which others have mooted, namely that the fire was arson, and (my refinement) that the fire then provided a handy cover/pretext for re-modelling the Shroud. Thus the massive burn holes could have been in selected places, e.g. the junction of the arms and shoulders, intended to obliterate aspects of the image that had met with scepticism. Note the convenient bilateral symmetry around the vertical midline, which required the Shroud was folded vertically through the middle of the face (!). What a way to treat an icon, a holy relic no less, even before flames and heat entered the equation. Needless to say, none of this invalidates what follows re the opportunities, missed or otherwise, (but mainly missed on account of the 2002 “restoration”) for gleaning unique and irreplaceable scientific information from bloodstains etc that had been partially incinerated for whatever reason – albeit under non-laboratory conditions – but merely to remind the reader to keep in mind the possibility that there may be more than meets the eye when discussing the 1532 fire and its immediate aftermath.
We would never use the same destructive methods of observation on an undamaged relic, but misadventure gave us many unexpected options.
Yes, why throw anything away? Why not bag it up, catalogue it, and make it available to researchers, present and future, in small instalments, as and when analytical methods become sensitive? The Vatican was quoted not so long ago as saying that the Shroud was “impossible to falsify”. No, it is not falsifiable if scientists are to be denied further access, even to burned bits deemed aesthetically unattractive, excised and then binned. Or will some of the bits re-emerge in years to come, having been released to selected scientists who can be relied upon to make claims that if not pro-authenticity at least are not anti-authenticity? (Such things are not unknown).
The important fact is that, before the restoration, we could look at the chemistry of specific locations on the Shroud where scorches intersected image, blood, serum, and water stains. The restoration destroyed much chemical information at those intersections.
Agreed … but do we have the techniques – even now – to probe the nature of a faint discoloration, allegedly a mere 200nm thick? Let’s exercise a little humility. The very best 20th century techniques revealed next to nothing about the image, except that it resembled a scorch (dehydrated carbohydrate etc.) reminiscent of scorching, reminiscent of pyrolysis needing only a raised temperature. But that was it – no confirmation, no decisive rejection. That then left the field wide open for Rogers to pull a white rabbit called Maillard out of his chemist’s hat, requiring a lot more guesswork and assumptions than plain old pyrolysis – like an impurity layer, starch fragments -a highly questionable proxy for reducing sugars (shame about the lack of aldehydic reducing groups), volatile putrefaction amines, orderly diffusion, if there is such a thing, or even orthogonal projection of collimated molecules – for which there is no such thing except in a vacuum with barriers and slits. So let’s not tear out too much hair on account of the 2002 “restoration” given the liberties that have been taken with the present data or lack thereof. The Shroud custodians can maybe be forgiven a less-than-enthusiastic response to calls for still more “science” given the shambles of what has happened so far when largely self-appointed scientists (“experts”) decide the time is ripe to apply all the latest in smart technology (ha ha) revealing next to nothing about the Shroud image, or the role of the linen vis-a-vis that largely imaginary “impurity layer”. Let’s not mince our words – STURP’s ‘shroudology’ has severely damaged the reputation of science, and continues to do so to this day. Is it any wonder the Shroud custodians are proving stubborn unimpressed and unhelpful less than helpful, especially as it was a STURP porphyrin specialist (Alan D Adler) who having morphed overnight into a textile conservation expert ‘successfully’ cajoled for the 2002 restoration – see below under ‘autocatalysis’. Some might be forgiven for thinking that the main danger to the Shroud has never been from reactive free radicals, but from ballooning egos and multidisciplinary one-man-bands – past and possibly present.
If the image had been painted or retouched, some foreign materials had to be added to the cloth. The pigments and vehicles (e.g., the ochers, realgar, orpiment, mosaic gold, glair, gums, and glues) would have been subjected to a violent “chemical test” during the fire. The temperatures, temperature gradients, pyrolysis products, and water used to extinguish the fire would have changed the chemical composition of most foreign materials. Before going to Turin in 1978, we did many experiments on the stability of the painting materials. We had hoped that future observations on the Shroud could compare predictions with reality. The restoration disturbed exactly the areas of most chemical importance.
Yes, the 2002 ‘restoration’ should never have happened without much wider discussion and consultation beforehand. It should have had an agreed protocol that insisted on conservation of all material, burned/scorched/semi-pyrolysed (call them what you want) fragments. The fact that it did not says there is something not right about the interface between religion and science. Well, that’s plain for all to see, at least for those who, like me, spend a lot of time (probably too much) on the internet. Part of the problem is the way in which many sceptical thinkers, scientists, philosophers etc have been labelled or written off as ‘atheist’ (I saw Barrie Schwortz dismiss Joe Nickell with that pejorative tag back in February I can’t speak for Joe Nickell, but I can for myself. To me, the term atheism does not mean without Jehovah, or God, or Jesus or Allah. It means “without theism” in all its multiplicity of forms with unique, distinctive but conflicting theologies – the cause of so much strife. Think about it… To me there is only one IMPORTANT atheism where the world of ideas is concerned : atheism in my book is, as I have said, simply “without theism”. God, if He/She/It exists, probably has no desire or need for Earthlings to be constantly at each others’ throats over differences in the way that He/She/It is perceived, or over supposed prescriptions and proscriptions from on high. End of alternative-sermon.
Returning at 15:00, 7th Jan. having just dealt with the theology question (a mere detail!)
The persons involved in the restoration of June and July 2002 did not appear to be familiar with previous scientific observations, and they did not consult chemists with different areas of experience or chemically-oriented textile conservators. The restoration destroyed much of the chemical information that could have been recovered as a function of position on the surface of the Shroud.
Yes, but did Ray Rogers ever consult sufficiently with “chemists with different areas of experience or chemically-oriented textile conservators”? “Consult ” can mean different things to different people (let’s not go into that).
The fire of 1532 produced many extremely reactive pyrolysis products, and the fire was extinguished with water.
Yes, but it’s questionable whether the “reactive pyrolysis products” were inside as well as outside the reliquary, or even “inside” (see earlier discussion). Nobody can say for certain how those burn holes, scorch marks etc were acquired, or, for that matter, where water enters the equation. (Yes, there are water marks on the Shroud, but as Hugh Farey pointed out recently, there was an influential paper (insert ref) that argues that the water marks and burn holes were acquired at different times).
All paints that were used during or before medieval times (except gold) are changed by heat and/or the chemically reducing and reactive pyrolysis products of the cloth (e.g., formaldehyde, furfural, organic acids, CO, etc.). For example, red hematite would have been reduced to black magnetite. This fact provided one basis for refuting McCrone’s claim that the image was painted with hematite.
How can one say that red haematite (Fe2O3 with hydration water) WOULD have been reduced to black haematite magnetite. Yes, we know that the latter, Fe3O4, has a slightly lower oxidation state (empirical formulae show the lowest, simplest ratios, but for accounting purposes they can be scaled up in this instance to Fe6O9 and Fe6O8 respectively, showing magnetite to be slightly less oxidised). But no one can say for certain that one was, or could be, definitely converted into the other, simply as a result of an incident in 1532.
(Technical note: red rust is Fe2O3, but the black gunge that forms in one’s CH radiators is Fe3O4 on account of a reducing environment, due to electrical galvanic action between steel radiator and copper pipe, forming hydrogen that has to be periodically bled off with the radiator key).
We planned to look for the products of such reactions. Some medieval painting materials become water soluble, and they would have moved with the water. A huge amount of chemical information existed in the scorches.
So why were scorches not looked at more closely in 1978? Why was there so much wild speculation about the location and nature of the image before all the relevant and available evidence was collected and analysed?
Most organic colors are much less stable than cellulose (linen) and the normal inorganic pigments. Experiments in 1978 showed that scorch lines in impurities precede the scorches in pure linen. Most organic materials, including natural products, change in predictable ways in response to heating and the known products of cellulose pyrolysis. We even tested squid ink, which had been reported being used in ancient times.
Scorch lines? Relevance of cellulose pyrolysis (which probably did not happen -something on which Rogers and I concur)? Meaning unclear, there being a surfeit of detail here aka chemist’s waffle, that is not justified by the little we know – or can ever know- regarding the precise circumstances and mechanism of the imaging process. So what we are looking at basically is … more waffle…
It might still be possible to extract the products of the reactions from the materials recovered during the restoration, assuming that samples were segregated and locations were recorded. Such information could be important for suggesting the chemical composition of the image.
Real information content: essentially zero. Not what one expects in a set of FAQs. No wonder this project is taking so long to complete. More waffle
Most possibilities for directly studying the effects of the fire on image materials were destroyed by the restoration of 2002.
Waffle.
Visual and microscopic observations on the Shroud in 1978 indicated that image color or its reaction products did not move with the water. Other unidentified products did move. Aldo Guerreschi has suggested that two different sets of water stains exist on the Shroud. They could contain interesting chemical and historical information. We had counted on the tape samples and possible future direct studies on the scorch/water-stain areas of the Shroud for detailed chemical confirmation of what did and did not move with the water. Now the tape samples are kept from scientific study by the officials in Turin, and scorches were destroyed by the “restoration.”
Still more waffle…
The Shroud showed many locations where scorches of different severity intersected image and/or blood. Thermal gradients can be estimated on the basis of scorch colors.
Waffle
Temperatures are the most important factors in calculating chemical rates. We made predictions on the kinds of products that might appear in image areas as a result of reactions between its components and the pyrolysis products and water. These predictions could be used to test many of the hypotheses that have been proposed for image formation.
Yes, well that’s a big point iof contention between me, a biochemist by training who occasionally was called upon to teach energetics in Chemistry (to London University A-Level) and Ray Rogers, a professional thermochemist. I know nothing about his pre-Shroud work, except that he used kinetic methods to establish the safety of chemical explosives after storage. (A lot of that work was classified, his being employed by the US defence industry at its Los Alamos research institute). What I do know is that his references to temperature in connection with the Shroud, e.g. to argue for a Maillard reaction at close to (recently deceased) body temperature, focus exclusively on chemical kinetics. Chemical thermodynamics NEVER get a look in. Elsewhere I have argued that Rogers’ proposed reaction between a volatile amine and a reducing sugar (even if both are present – which is highly conjectural) is improbable not just on kinetic but thermodynamic grounds. The reason why a reaction is unwilling to go at a low temperature , even 40 degrees C, might NOT just because of kinetic factors to do the frequency of particle collisions, or the energy of collisions (“kinetics). It can because the reaction is simply n0t feasible at that temperature, because the overall entropy/free energy change is unfavourable to reaction. Raising the temperature does not just increase the rate of reaction. It can endow the products or environment with greater entropy (disorder, chaos, more arrangements) making a reverse reaction less likely, giving directionality to the reaction, making it “go”…
I find it extraordinary that Rogers, a founding and managing editor of Thermochmica Acta (in which he published his controversial attempts to dismiss the radiocarbon dating) never once, to the best of knowledge, made any reference to thermodynamics when attempting to argue for the feasibility of a Maillard reaction betwen gaseous amines and starch fragments at temperatures. According to Rogers the latter was entirely feasible just a few degrees above normal body temperature (post mortem thermogenesis, you know, or maybe didn’t) while the literature on melanoidins and Maillard reactions constantly stresses the need for much higher temperature (typically 150 degrees C and higher) . Bread in a faulty toaster does not brown slowly at 100 degrees C – it does not brown at all. That’s nothing to do with chemical kinetics, and everything to do with thermodynamics. Ripe ammonia -releasing Brie or Camambert that is brought to room temperature close to bread does not cause the bread to go brown… despite the reducing sugar.
More to come:
Logging off at 16:35. Still more to come under FAQ 15 (sigh). Back tomorrow (Tuesday) hopefully to complete this hugely unrewarding task.
Resuming at 10:42 Tuesday 8th Jan
I took samples from many scorch/water/image intersections in 1978, but observations on them generated more questions. Answers required additional observations and/or samples. The samples are now secreted in Turin. As a result of the restoration, any future studies will be much more difficult and expensive: Some will be impossible.
Has anyone asked the custodians for an explanation?
The Shroud is a structure composed of chemical compounds, and all of the main ones have been studied in detail. They are published in chemical text books. Chemical analyses can yield considerable definitive historical information. All manipulations of the Shroud should be considered in detail in order to preserve as much information as possible.
Yawn…
Linen-production technology has changed through the centuries. We have assembled chemical information related to the technology, and we have consulted textile experts who have done detailed chemical research that relates to the composition of the Shroud. Our detailed analyses suggested that the cloth had been prepared by technology common before about AD 1200. It best resembles linen made in the Near East during Roman times.
Where’s the detailed analysis to support that claim? Detailed analyses for the alleged 1st AD processing aids (starch, saponins)?.Methinks the starch and saponins, in amounts sufficient to serve as an even image-capturing layer, were largely a figment of Mr.Rogers’ imagination
These results do not agree with the date published in 1989. The differences can be explained on the basis of samples from the radiocarbon area, but all scientific observations should be confirmed. Samples from the restoration might help confirm the properties of the radiocarbon sample; however, the persons involved in the restoration fight any attempt to test and confirm the truth. No scientist in Torino will discuss the problem, and the custodians refuse to recognize the problem. Ethical science is impossible in such an environment.
OK, but this is getting somewhat tedious and repetitive, and is hardly what one expects to find in a list of FAQs. What we see here are repeated gripes.
Lignin is a structural polymer that is found in all plants, including flax. Linen is bleached in an effort to remove as much lignin as possible, but some lignin always remains in linen. Lignin slowly ages with the loss of vanillin (4-hydroxy-2-methoxybenzaldehyde). A very sensitive microchemical test exists for the detection of traces of vanillin. It is easy to detect vanillin in modern lignin, it is harder to find in Medieval linen, and no test can be obtained from the few Shroud fibers that are still available for study. The lignin in samples from the Dead Sea scrolls (ca. AD 70) does not give the vanillin test. This observation would suggest that the linen of the Shroud is very old, casting doubt on the accuracy of the 1988 date. Observations on the lignin could be confirmed with samples from the “restoration”; however, such samples are jealously guarded in Turin.
The appropriate response to objections to the way the Shroud was sampled was to have agitated to have the entire procedure repeated with a better sampling frame. Replacing good physics (radiochemical decay) with bad chemistry (lignin degradation) was not the answer. (Oh, and on a technical note, one that was raised some years ago by Steven Schafersman, why was Rogers content with a quickie colour spot colour tests for vanillin? How quantitative was that? Why did he not use his mass-spectrometer to get quantitative data? Peculiar…
The tape samples show that much of the charred material is elemental carbon. It is very inert chemically. It would not have changed during the 470 years since the fire. Published concerns about isotope fractionation during the fire are nonsense. The carbonized material can easily be chemically cleaned of any organic deposits that might have appeared after the fire, making it an ideal material for radiocarbon dating. Before the restoration, the carbon from specific areas could have been dated separately, giving critical information about the homogeneity of the cloth as well as “clusters” of dates. Clusters of dates are more reliable than dates on single samples.
Agreed.
More to follow (there seems no end to this FAQ 15 …)
Dr. Max Frei took tape samples to recover pollen grains from the surface of the Shroud in 1973 and 1978. Sweeping claims have been made on the basis of Frei’s samples, but published photomicrographs do not support the claims. Other reports suggest that there were major changes in the number of grains found on Frei’s tapes between the time of his death and more recent publications. The pollen data badly need confirmation. The restoration totally destroyed any chance to take valid additional pollen-grain samples from the surface of the Shroud. A suspicious person might wonder whether the “restoration” was rushed through to prevent ethical work on confirming both chemical and pollen observations.
Noted. No comment.
Biblical accounts suggested several types of compounds that might have appeared on the cloth (e.g., aloes, myrrh, sebaceous secretions, etc.). We planned and executed chemical analytical methods that could detect them in 1978. Those methods were extremely sensitive, but they did not detect squalene or myrrh. These results could have been confirmed by additional tests on the Shroud, but the “restoration” has totally changed the Shroud’s surface.
Shame. But am I the only one to suspect that there’s now developing more than a hint of scapegoat-hunting here (for Ray Rogers’ and STURP’s inability to provide any kind of definitive answers despite some 20 or 25 years of investigation)? Needing access to, and being deprived those burned bits in 2002 would seem to be clutching at straws…
The surface of the Shroud could have been analyzed by Electron Spectroscopy for Chemical Analysis (ESCA), which observes the top few nanometers of the surface. Now that the surface has been disturbed, that powerful technique will be much more difficult to apply, and results will be ambiguous. This is a terrible, discouraging loss for Shroud chemists.
Agreed. You just can’t get the custodians these days…
The problems associated with surface analyses are now compounded by the fact that thymol was used to sterilize the reliquary after the 1988 sampling operation. Thymol is a phenolic compound that will react with many functional groups on the Shroud. This will confuse image analyses, and it may result in damage to the cloth. As one example, we found a significant amount of iron in the Shroud’s cloth. Iron reacts with phenolic compounds to form complexes, and some of them are intensely colored. I would urge the custodians of the Shroud to consult with chemists before taking other irreversible actions.
Agreed.
One justification for the hurried, secretive restoration was a fear of “autocatalytic” degradation of the cloth. No experts on chemical kinetics were consulted. The Shroud has not been and is not now in danger of autocatalytic degradation (see FAQ 6).
Whether autocatalysis represents a credible risk is not for me to say. What i do know is that the phenomenon involves a lot more than chemical kinetics, and I doubt whether it can be ruled out so categorically purely on kinetic grounds.
What is clear is that Alan Adler, porphyrin specialist was consulted, and Ray Rogers, thermochemist/chemical kineticist was not. One suspects this was as much about egos as it was about science.
Chemical autocatalysis is responsible for the destruction of books that are made with cheap, acid paper. Claiming analogy with the Shroud is mischievous. Adler and Schwalbe made the following comment: “Previous chemical reactions on the cloth, e.g., the retting process in manufacture of the linen, the known historic fire and its extinguishment, and previous display and storage procedures, have left a variety of chemical structures on the surface that can act as oxidants and also as catalysts. For example, the acidic structures produced by previous oxidative activity can strongly promote various types of autocatalysis” [A. D. Adler and L. A. Schwalbe, “Conservation of the Shroud of Turin,” Shroud Spectrum International, No. 42, December 1993, Indiana Center for Shroud Studies].
Such claims led to the secret restoration. Secrecy is never productive, and the plans for a restoration should have been reviewed with as large a group of scientists as possible. The restoration was a terrible mistake.
Amen. And that, finally, is the end of FAQ 15. For a while, I feared we were stuck in an endless loop…
……………………………………….
“See FAQ 15”
Response: So there are 18 FAQs, not 19 as per numbered list, given that two have been combined into one.
Well, that’s a bit less work for me to do (except that FAQ 15 appears to go on and on and on…sigh). So please scroll UP to see FAQ 15, given I am posting in reverse chronological order.
Archaeologists use many different methods to estimate the age of artifacts and/or soil strata that contain artifacts. One of the most important ways is to observe changes in technology: methods used to make tools change with time. There is a big difference between the hand axes made during the Paleolithic and fine arrow points made a few hundred years ago. The technology used to make the Shroud was much different than that used during medieval times or modern times.
””””””””””””””’
Response: It was I who applied bold format to that last sentence. It really tells one all one needs to know about Rogers’ objectivity – or lack thereof. Ray Rogers clearly joined the STURP team (by invitation I believe) convinced of the Shroud’s authenticity, and hoping to prove its authenticity by scientific means (he was not to know that radiocarbon dating would later result in a medieval date, a result that he so vehemently attempted to undermine with his home-based microscopy of an left-over thread or two acquired through “contacts”)
It’s a moot point as to whether a scientist can be said to have abandoned objectivity by taking a prior position, as clearly Rogers did. If the answer is yes, then lots of scientists can stand accused of being less-than-objective. But it’s the human condition to have hunches, and to have those hunches colour one’s perceptions.
I’m still not certain of the answer, despite a career in science and teaching. As I said in a recent post, science performs best when there is an overlap between fact-finding and speculation – aka model building. One could be perfectionist and suggest that every individual scientist must find the perfect overlap between those two, and stay in that overlap area of the Venn diagram. But science is a communal activity, so one could argue that there is no need for every scientist to be so saint-like, with a self-denying ordinance to avoid operating outside of the present body of knowledge or theory(which may be paltry) to avoid being seen as less than saint like. Well, I for one never pretended for a moment when I entered science research that I had to be saint- like. In the final analysis, science is self-correcting, or, put another way, one’s sins, one’s errors of commission or omission will sooner or later find one out.
What matters is not what WAS in the head of the scientist when he or she comes to write their report for publication. What matters is the ability to stand aloof from one’s own previous perceptions, and attempt to write the paper as if were someone else’s result, and to be as honest and self-critical as possible – without going too far as to destroy one’s own credibility.
Had I been Rogers, writing these FAQs, I would not have written that bolded sentence ( The technology used to make the Shroud was much different than that used during medieval times or modern times.). It raises too many questions, places too many doubts in the reader’s mind, as to the writer’s objectivity. I doubt whether that sentence would ever have been permitted by a referee for any prestigious peer-reviewed journal (with the possible exception of Thermochimica Acta – see my earlier comment)
Stone tools hydrate and form a patina. Its thickness indicates age. Similarly, all organic materials tend to decompose or change structure with time. Proteins undergo “racemization.” Their amino acids change their optical properties. This would apply to the blood on the Shroud.
The DNA in blood and tissue samples degrades with time. The DNA in Shroud blood samples shows the effects of significant aging: only short lengths of the chain remain intact. The reported ABO typing results are very suspect and probably not valid. However, the results prove appreciable age for the Shroud.
Crystalline materials undergo damage that is caused by natural sources of radiation, and Shroud fibers show some evidence for changes in their crystal structure.
Some compounds like lignin change composition with time. The lignin in the Shroud does not give the normal microchemical test for vanillin, indicating that it is quite old. Measurements of the chemical rate for loss of vanillin estimates an age for the Shroud of more than 1300 years, depending on storage conditions.
Response: It is hard to take serious issue with the long preamble, except for that final carefully and lovingly “take-away” message (pure Rogers), which I have bolded. Evidence from the degradation of a chemical marker for so long a time period, whether 750 years or longer, and influenced by so many unknown environmental factors, is at best suggestive. What we see here is not science, certainly not QUANTITATIVE science. It is, if the truth be told, little more than self-promoting PR. It is arguably not the job of a research scientist to write oracle-posturing FAQs. Ray Rogers should have stayed in the background, maybe as one of several consultants. It’s yet another instance, as if any more were needed, of Raymond Rogers performing, or attempting to perform, as a one-man-band. That sort of thing is best left, in my opinion, to seaside piers.
Because the cellulose was not colored, the impurities had to be significantly less stable than cellulose. This also suggested that the impurities were the result of cloth-production methods, and they should appear on all parts of the cloth.
Response: still showing that blind spot for the PCW…
A search for carbohydrate impurities on the Shroud confirmed McCrone’s detection of some starch fractions.
Response. Why the fixation with starch where image is imprinting is concerned? Where in the literature do we find starch being suggested as a medium par excellence for accepting an image? OK, so it might be more easily ‘dehydratable’ (pyrolysed? scorched? caramelised?) than the cellulose of the secondary cell wall, as Rogers has previoulsy suggested, being less crystalline, but might equally well be less cooperative than the carbohydrates of the primary cell wall, notably the hemicelluloses and other pentosans.
Why no decent quantitative or eeven qualitative data on starch content and distribution on the Shroud, or relative amounts in image v non-image area. Why base so radical a theory as amine-reducing sugar interaction i.e. (Maillard reaction) on starch (which is non-reducing unless extensively degraded to almost sugar-like dimensions) on almost non-existent analytical data, being forced to rely on others’ findings of “trace amounts”, and/or detected by an unsuitable reagent designed for an entirely different test (I refer to the red-colour seen with an iodine/azide reagent that was designed to test for sulphur-containing proteins, instead of iodine/potassium iodide which is conventional for starch detection).
Rogers has failed to convince there is/was sufficient starch on the Shroud to give ay kind of n image, regardless of mechanism, lacking as he did high grade analytical data. In any case, his proposed mechanism (Maillard reaction) needed simple reducing sugars – not starch or ill-defined “starch fragments”. Pseudoscience.
Starch and low-molecular-weight carbohydrates from crude starch would color much more easily than would cellulose as a result of either thermal dehydration or chemical reactions.
Response: Yes, but as said above, the relevant comparison is between starch and PCW hemicelluloses, not starch and secondary cell wall celluloses. And it should be reducing sugars that are discussed in the context of Maillard reactions, not starch. More pseudoscience.
Any image-formation mechanism that would result in color formation inside the linen fibers must be rejected.
Response: Yes. It’s not the SCW that needs to be discussed, but the more superficial PCW.
Some “theories” that have been mentioned that would cause coloration inside fibers are penetrating radiation, high temperature scorching (hot statue, painting with a torch, etc.), and catalyzed dehydration of the cellulose. Image fibers are colored only on their surfaces.
Response: This is getting monotonous. Tunnel vision. Blind spot. if not pseudo-, at least misdirected science…
Prof. Alan Adler of Western Connecticut University found that the image color could be reduced with a diimide reagent, leaving colorless, undamaged linen fibers behind.
Response: Adler’s finding with the diimide (HN=NH) – a powerful reducing/hydrogenating agent was an important one. It provides one of the few clues as to the nature of the Shroud image. As mentioned earlier (below) it indicates that the Shroud image owes its yellow colour to one or more double bonds in superficially modified linen, e.g C=C- . Technical note: there usually has to be a sequence of two or more conjugated (alternating single and double bond) to confer an appreciable yellow colour when viewed under ordinary white light, producing a delocalised system of laterally-overlapping pi-bond orbitals, as would exist in, say, -C-C=C-C=C- etc. What diimide presumably does is to convert one or more of those double bonds to single bonds, interrupting the conjugation, e.g. by making -C-CH-CH-C=C- as a first step (maybe the second would be hydrogenated, but one hydrogenated bond might be sufficient to produce bleaching). So where does the oft-cited “dehydration” enter the equation, so to speak?. It’s the presumed initial step that generates the double bonds and coloration in the first instance. Polysaccharides (polymeric carbohydrates) have lots of hydroxylated carbon atoms which, when chemically dehydrated (loss of 2H atoms for each O atom) creates a C=C double bond.
That’s the chemistry, well established in the case of model systems for pyrolysis, i.e. thermal degradation without air or oxygen or anything else for that matter needing to be present (though likely to affect the outcome if present). But the statement you read above goes way beyond the established facts, preparing the ground for the speculative and contentious scenario-creation that follows in its wake. Let’s see why …
First let’s be absolutely clear as to the logic which we are being asked to accept. It starts with a yellow coloration on linen, which is woven from processed fibres. It says that a particular chemical, diimide, can bleach that colour. It then says that the bleached fibres now look as if they were completely as they were initially – i.e. colorless and “undamaged”. Here’s the punch line – the events that produced the initial colour , and the subsequent bleachig thereof, could not have occurred on the fibres themselves. They must have occurred on something else, something that was even more superficial than the most exposed fibre in linen. Ipso facto, it must have occurred on soemthing that was not a fibre , derived from the original flax plant. It must have occurred on something acquired, subsequent to the harvesting, retting and processing of flax fibre, in other words an extraneous , foreign non-indigenous , contaminating IMPURITY LAYER.
Can you spot the flaw in that logic? It’s maybe subtle, maybe not. But it’s a flaw all the same, and a serious one in view of the edifice that has been constructed on the foundations of that imputed “impurity layer”. The flaw is to imagine that because something looks “undamaged” after a cycle of chemical steps – coloration and bleaching – that there has been no change to that surface at the atomic or molecular level. That, dear reader, is one of the most egregious examples of chemical illiteracy that I have ever read from someone who is/was hailed as a top notch chemist.
Here’s the first analogy that comes to mind (I may be able to think of better ones – closer to carbohydrate chemistry later). Immediately after removal from a smelter, an aluminium ingot looks shiny. One would think the surface is pure aluminium metal. But it’s not. As soon as it encounters air and oxygen, it acquires a thin but highly coherent film of oxide that is reckoned to be some 50nm thick. That film is too thin to affect the appearance of the metal – it is shiny- but totally transforms the chemistry of aluminium. It protects the aluminium from further oxidation and corrosion. Without it, aluminium, high in the reactivity series (and thus difficult to manufacture except by electrolysis) would corrode away within hours, minutes even, when exposed to air. There is a simple way of demostrating that: rub a little mercury or mercury salt onto alumium. Within minutes the metal develops white whiskers of alumium oxide, becomes hot and quickly collapses to a white powder. Why? Because the mercury has lifted off the protective film of alumium oxide, allowing oxygen to get underneath and continue attacking thje metal unhindered.
The surface of any aluminium metal you meet in everyday life is “damaged” – it has a thin film of oxide – but you would not know that just by looking at it. But Rogers has assumed that because diimide-bleached Shroud linen looks undamaged, then it must also be undamaged at the atomic and molecular level. Based on that faulty conclusion, he has ruled out any change to the intrinsic fibres of linen, and set off on a wild goose chase in pursuit of surface “impurities”, quoting (or misquoting) ancient sources for ideas on what those impurities might be (starch, starch fragments, saponins etc). That goose chase continue to this day, so much so that it seems that it has never occurred to some folk that the image might be on the most superficial layer of the linen fibre – namely the PRIMARY CELL WALL (PCW). You will look in vain for any reference by Rogers to the PCW. It was not on his radar screen. He seemed to have a blind spot for the PCW, even when making occasional reference to the hemicelluloses of linen, and their greater chemical reactivity compared with ordered cellulose fibres. He did not seem to appreciate that those same reactive hemicelluloses are a major component of the superficial PCW, along with relatively disordered cellulose.
One can be the most brilliant chemist in the world, but that counts for little if one is working on a linen artefact, and fails to appreciate its botanical microstructure. Rogers seemed to have little interest, and even less understanding of the botanical microstructure of flax fibres at the cellular level. He seemed to perceive them merely as a mix of chemical components, and concentrating on the one that is quantitatively the most important (cellulose), and failing to realize that what he assumed to be a coating of impurities that could be reversibly coloured or bleached was in all probability the PRIMARY CELL WALL of the flax and linen fibre. Thus was “Shroudology” led into a cul-de-sac from which for many there has been no escape, back to the world of botanical and chemical reality.
I have been criticized elsewhere for choosing a “cumbersome” title for this posting. Be that as it may, those few words summarize this entire posting. By putting that title into cyberspace, gradually to be picked up by search engines and circulated I hope in the fullness of time to undo some of the damage inflicted on “Shroudology” by someone well-versed in chemistry, but with a disastrous deficit in his knowledge of botany. Shroudology needs to be set back firmly on track. The means for doing that can be discussed on another occasion. Maybe there’s a role for those surface microprobes that are proposed by a certain Spanish gentleman whose name I have mislaid (insert later). The first task could be to examine Shroud fibres before and after diimide treatment and see if they really are “undamaged” as claimed (or assumed) by Raymond Rogers – STURP’s chemical team leader.
I’ve expended a lot of time and words on that one sentence above, bit considered it essential, in view of that “edifice” that is constructed on the basis of a flaw in logic, or at any rate, chemical logic.
Here’s the edifice that followed:
This confirmed spectral data that indicated that the image color was a result of complex conjugated double bonds; however, it proved that image color was found only on the outer surfaces of colored image fibers. Until this time, we had assumed that the image color was a result of chemical changes in the cellulose of the linen. The most likely change would involve the dehydration of the cellulose to produce conjugated-double-bond systems Adler’s observations proved that the cellulose was not involved in image formation. This is an extremely important observation.
This fact was confirmed by the observation that the image color on some fibers had been stripped off of their surfaces by the adhesive of the sampling tapes. The photomicrograph shows the places where two fibers were pulled out of the adhesive leaving their colored coating behind. The coating is too thin to measure accurately with a standard microscope; however, it appears to be 200-600 nanometers thick (in the range of a wavelength of visible light).
The bands of color and the fact that all of the image color appears only on the outer surfaces of the fibers, suggested that image formation involved a thin layer of impurities.
This also suggested that the impurities were the result of cloth-production methods, and they should appear on all parts of the cloth. A search for carbohydrate impurities on the Shroud confirmed McCrone’s detection of some starch fractions. Starch and low-molecular-weight carbohydrates from crude starch would color much more easily than would cellulose as a result of either thermal dehydration or chemical reactions.
Any image-formation mechanism that would result in color formation inside the linen fibers must be rejected. Some “theories” that have been mentioned that would cause coloration inside fibers are penetrating radiation, high temperature scorching (hot statue, painting with a torch, etc.), and catalyzed dehydration of the cellulose. Image fibers are colored only on their surfaces.
Response: What can I say that I have not said already? In common internet parlance, having “dug a hole for himself”, Ray Rogers just continued digging…
Break off at 14:20 Sunday. More later.
The University of Tennessee maintains an experimental area where observations are made on decomposing corpses. They find that flies lay their eggs in wounds on dead bodies, and maggots appear before 30 hours at about 23ºC. This approximates the time required for liquid decomposition products to begin to appear on the surface of a body. We could not find any evidence for the migration of liquid decomposition products through the cloth; therefore, the cloth could not have been in contact with the body for very long.
Response, last updated at 10:45 today (Sunday). More to follow.
Extraordinary! Bizarre even! Rogers introduces the topics of “decomposing corpses” and then immediately launches into this strong stuff about blow flies and maggots – a grim reality to forensic pathologists maybe when a body has been discovered in woodland by a dog-walker after lying there several days and nights at the height of summer. But that is all of somewhat dubious relevance to the Crucifixion and what followed, given the relatively shorter time scales, and given the immediate interest and concern evident from the Biblical account in the respectful disposal of the victim, with no time wasted in protecting the corpse from flying insects (which I assume, rightly or wrongly, to be what the reference to “100 lbs of aloes and myrrh” was all about, assuming it was a true account, and not a later cosmetic tacked-on addition.)
More to follow under this FAQ 11 heading…
Decomposing bodies start producing ammonia (NH3) in the lungs quite soon after death, and the ammonia diffuses outward through the nose and mouth. Ammonia is lighter than air, and it diffuses rapidly. The rate of production of ammonia decreases with time after death.
Response: this may or may not be true (that final sentence makes no sense to me whatsoever, unless there is a temporary hiatus in ammonia production that has been mistaken for complete cessation). In any case, this kind of information has no predictive utility unless numbers are attached. Rogers however is showing here a total lack of concern for precise quantitation – and in pushing his ‘incipient putrefaction’ scenario to account for the Shroud image in so premature and precipitate a fashion was abandoning in a cavalier fashion the distinctive modus operandi of the true scientist, while never being backward in condemning others for exhibiting bias or taking short cuts. Yes, Rogers frequently charged others with ‘pseudoscience’ – yet here we see precisely that. OK, so he later did some experiments ( with Anna Arnoldi as I recall) presenting some evidence in favour of imaging onto dextrins by gaseous ammonia in a simple model system- the details of which I questioned some months ago (must track down reference) – but it is this frequently cited list of FAQs that is in the frame right now – the ‘user-friendly’ account intended for non-science specialists – the kind of “press release” that is intended to mould opinion. I make no secret of my distaste for the promulgation and non peer-reviewed science in this fashion. And before anyone reminds me that some of Rogers’ work , notably his critique of the radiocarbon dating based on mysteriously-acquired threads, appeared in that prestigious journal, Thermochimica Acta, I suggest readers look up the wiki entry for Raymond N Rogers, and note Rogers’ involvement in the creation and early management of that journal.
Here’s the relevant passage: “He was also on the editorial board of Thermochimica Acta from the first issue of this journal in 1970 (also the very first paper published in the first issue of this journal is authored by him) until his retirement in 1988.”
(Nope, there’s now’t wrong with a journal founder or managing editor publishing in “his own” journal, provided that folk know that to be the case*especially where matters of public controversy are concerned, and where the research might be considered to be of dubious relevance to the journal’s mainstream interests , escaping the closer critical scrutiny that would be accorded to more conventional submissions.)
* noting that so well-informed a scientist (and critic of Ray Rogers) as Dr. Steven Schafersman seemed unaware of the Thermochimica Acta connection when criticizing that journal’s editor for having accepted for publication Rogers’ evidence against the radiocarbon dating.
Within a few hours, depending on weather conditions, a body starts to produce heavier amines in its tissues, e.g., putrescine (1,4-diaminobutane), and cadaverine (1,5-diaminopentane). These amines are much heavier than air, and they diffuse relatively slowly.
This is not just vague (and potentially misleading) in a quantitative sense (no numbers). It’s also vague in a qualitative sense, and may raise doubts even in the minds of non-scientists that Rogers is telling it the way it is. After all, there are lay folk who have experience of hanging game ( venison, pheasants etc) for days or even weeks one end, and while being familiar with the ‘gamey’ aroma will not have noticed a pungent ammonia aroma, as with that from over-ripe Camembert – and certainly not a stomach-retching aroma from higher amines like those listed.
Post-mortem putrefaction is not a topic I am especially keen to get into right now. Suffice it to say that there are crucial details that have to be considered, notably the nature of the gut contents at the time of death, especially the proteins, given that it is probably the colonic microflora with their bacteria, and the enzymes associated with those bacteria (amine-forming amino acid decarboxylases especially) acting on those dietary proteins that play a major role where emission of gaseous amines and/or liquid exudates is concerned – at least in the short to medium term (hours and days, rather than weeks).
Response: Yes, a chemist with mass spectrometer might well be able to detect traces or putrescine etc within hours of death. Whether the human nose would do so (or flying insects) is another matter, if the initial output was small. To attempt to envisage and construct an image-imprinting mechanism on so tendentious a scenario – occuring a mere day or two after death under so many noses – does not strike me as terribly good science. Indeed, when embroidered as we see here with a selection of the kind of ‘techie’ allusions and asides that we see above (relative diffusion rates etc) then it smacks of that very thing that Rogers himself so abhorred – or professed to do so – namely pseudoscience.
Experiments prove that slow diffusion relates to increased resolution in image formation.
Response: Pseudoscience, masquerading as science…
The early appearance and rapid diffusion of low-molecular-weight ammonia from the nose and mouth might help explain the greater amount of image color between the nose and mouth, in the beard, and into the nearby hair.
So that ammonia from the lungs (? or gut via the oesophagus?) gives a “greater” amount of image colour around the nose and mouth. What if there had been no supplementary ammonia? Are we to understand that there is background ammonia from every pore of the skin that would produce an image, albeit fainter, without the extra? Is a recently deceased corpse really equivalent to Camembert cheese?
Oh, and is the prominent imaging of the hair really due to ammonia that has made a side trip to that location. Yes, I know that it is difficult to explain the imaging of hair from ammonia without invoking that extra source, given that hair is keratin that does not putrefy, and given that hair would tend to trap any ammonia issuing from the scalp.
This is all silly beyond words. But for the context, it would be risible. Sadly, it is pseudoscience, unworthy of a seasoned chemist.
It will also diffuse through the cloth more quickly and reach the back side of the cloth in greater concentration. Ammonia will diffuse about 20 cm through air while cadaverine is diffusing only 6 cm.
Now we see Rogers really getting into his stride, with the implicit assumption that there are chemically-reactive amines galore issuing from a corpse and rapidly getting to where they are needed to perform their image-imprinting function. Shame then that there are no well-attested practical grounds for thinking that a stream of gaseous molecules ever projected a recognizable image of a nearby object across an air gap onto a sheet of linen. Diffusing molecules do not behave like etching laser beams. They spread, due to their intrinsic kinetic energy, resulting in millions of random collisions with neighbours that instantly deflect them from their initial course or direction. It’s hardly laminar flow, is it, mapping a replica at a distance. It’s hardly na air blade hand-drier and fax machine, rolled into one, is it? Neitherof those two has to rely (as a first step) on natural heat convection to produce orthogonal projection.
Oh dear, oh dear, oh dear. What a way to end a career, to lose the plot in so spectacular a fashion.
I’ve heard of white rabbits being pulled from of a hat. What we see here is no white rabbit. It is long-deceased road-kill, the victim of a chemist in a hurry, travelling at indecent speed, leaving behind a result that is unpleasant to deal with except with a clothes peg over one’s nose.
 When a cloth is dried on a line, impurities concentrate on both evaporating surfaces; however, more impurities will deposit on whichever surface dries faster. Any concentration of impurities can take part in the image-formation reactions. This can explain the “doubly superficial” image.
When a cloth is dried on a line, impurities concentrate on both evaporating surfaces; however, more impurities will deposit on whichever surface dries faster. Any concentration of impurities can take part in the image-formation reactions. This can explain the “doubly superficial” image.
Response: The “doubly-superficial” image needs to be explained and qualified. Only a few regions of the Man on the TS image appear on the obverse side of the frontal image – it’s mainly the face and part of hands as I recall. They can be explained in other models, notably scorching from a template, by virtue of being prominent 3D features lying above the general plane that make a greater indentation in linen, especially if there was applied pressure between linen and template.
OK, so the extrinsic impurity theory can explain it. But that is assuming the extrinsic impurities were truly mobile solutes soluble, e.g. like the blue dye in Rogers’ model system, extendible in principle to low molecular weight“reducing sugars” needed for the proposed Maillard reaction. But the closest that Rogers came to explaining where reducing sugars come from was “starch” or “starch fragments”. The first of these, even if gelatinized starch (60 degrees C and higher) is still macromolecular and thus not truly soluble. It’s what’s described as a colloid – dispersed rather than dissolved – and nowhere near as mobile or prone to rapid unhindered migration. But that’s the least of one’s objections to the extrinsic impurity theory – one that instantly blunts Occam’s Razor due to a veritable jungle of qualifying assumptions.
 Latest addition: 17:55 Because the cellulose was not involved in image formation, the color must have formed in impurities on the surfaces of the image fibers.
Latest addition: 17:55 Because the cellulose was not involved in image formation, the color must have formed in impurities on the surfaces of the image fibers.
My response: Nope. A non-sequitur of mind-boggling proportions. Cellulose as we have said is not the only carbohydrate intrinsic to flax or linen fibres. What we see here is what Chairman Mao might have called “The Great Leap Backwards”.
Independent observations have proved that all of the image color resides in a very thin layer on the outside surfaces of colored fibers.
Response: At last something we all agree on.
Evaporation concentration can explain the superficial nature of the image and the identical properties of the front and back images. It can also explain the “doubly-superficial” image, i.e., the presence of a superficial image on the back surface of the cloth as reported by Ghiberti and Fanti and Maggiolo.
Response: It could, but only if one was wedded to an impurity hypothesis and struggling to account for superficiality.
When a solution evaporates at the surface of a porous solid, dissolved solutes are concentrated at the evaporating surface. The principle is illustrated in the photomicrograph with blue dye. A piece of linen was saturated with a dilute solution of blue dye, and the cloth was dried while laying on a sheet of Teflon. All evaporation occurred at the top surface, and the dye concentrated on that surface. It is obvious that most of the dye deposited on the highest parts of the weave and the upward-pointing fibers of the nap. A sheet of cloth that contained sugars and starches would deposit those impurities at the very topmost part of the weave after washing and drying.
As above. (Sorry Rogers-disciples, if I seem harsh on your sadly-deceased guru. But it is you as much as Rogers himself who have prompted this critique. One tires of all the brow-beating, arm-waving, the impassioned megaphone advocacy and – most of all- the tunnel-vision. This retired science bod prefers to deal in cold hard facts and sensible non-emotive interpretation thereof.)
J. L. Banyasz, S. Li, J. Lyons-Hart, and K. H. Shafer [Fuel 80 (2001) 1757-1763] studied real-time evolution of formaldehyde, hydroxyacetaldehyde, CO, and CO2 from pure microcrystalline cellulose by EGA/FTIR (effluent gas analysis and Fourier transform infrared spectrometry). They detected 10 compounds simultaneously in the gas phase by FTIR. The cellulose decomposition is very complex. The quantity of formaldehyde produced is a function of heating rate, so decomposition mechanisms change depending on how fast you heat the cellulose. That is important in considering image-formation mechanisms and long-term stability vis-à-vis scorching processes.
According to A. G. W. Bradbury, Y. Sakai, and F. Shafizadch, [J. Appl. Polym. Sci. (1979) 23, pp. 3271-3280], the induction process in cellulose can be neglected above 300ºC. They observed two major decomposition mechanisms with the following constants:
E1 = 47.3 kcal/mole Z1 = 3.2 X 1014 s-1
E2 = 36.6 kcal/mole Z2 = 1.3 X 1010 s-1
They assumed that 65% of the products in the char-forming chain of reactions went to gas.
Glucose decomposes by a multi-step process. As with all of the other saccharides, the first is a dehydration/condensation reaction. Glucose decomposes by a multi-step process. As with all of the other saccharides, the first is a dehydration/condensation reaction. The condensation processes yield carbon-carbon double bonds, which ultimately lead to color formation. Bruce Waymack of Philip Morris measured the kinetics of the first reaction as E = 23.9 kcal/mole and Z = 1.26
Response (latest addition,17:35) : So, we are back with cellulose again, reinforcing the suspicion (nay, near certainty) that Rogers viewed cellulose as the only intrinsic component of linen on which an image might be imprinted. Having rejected cellulose – on grounds of its chemical stability – he then cast around for something else that he thought might substitute for cellulose – and settled for those extraneous impurities (starch, starch fragments, saponins .
Oh dear, oh dear, oh dear …. So let’s not tarry too long, given this just 8 out of 19 bullet points in Rogers’ so-called FAQ’s. (FAQs? As I said earlier, FQAs more like it – Frequently Questioned Answers).
“Results of kinetics studies support a low temperature image-formation process. The temperature was not high enough to change cellulose within the time available for image formation, and no char was produced.”
The findings with sticky tape – easy strippability of the image layer – should surely have ruled out cellulose pyrolysis – at least that part of the cellulose – close on 99% or greater – that resides in the thick secondary cell wall. But Rogers, as said before, seemed to have little or no concept of primary v secondary cell walls. Shame he did not (apparently) consult a botanist before promoting his extrinsic carbohydrate (“starch/saponin” impurity hypothesis.).
Added at: 16:50
Bands of slightly different color can be seen in Shroud photographs. They are most visible in ultraviolet-fluorescence photographs (see Hands UV).
Both warp and weft yarns show this property. Some areas show darker warp yarns and some show darker weft yarns. In some places bands of darker color cross. In other places bands of lighter color cross. The effect is somewhat like a plaid.
All of the bleaching processes used through history remove lignin and most associated flax impurities (e.g., flax wax and hemicelluloses). The more quantitative the bleaching process the whiter the product. The bands of different color on the Shroud are the end result of different amounts of impurities left from the bleaching process.
Anna Maria Donadoni, a curator at the Museum of Egyptology in Turin, pointed out locations where batches of yarn ended in the weave and new yarn had been inserted in order to continue weaving. The yarn ends were laid side by side, and the weave was compressed with the comb. The ends are often visible, and the overlaps correspond to zones of different color in the weave. The different batches of yarn show different colors.
Where darker bands of yarn intersect image areas, the image is darker. Where lighter bands intersect an image area, the image appears lighter. This proves that the image color is not a result of reactions in the cellulose of the linen. Some impurities on the surface of the different batches of yarn produced the image color. This observation is extremely important when tests are being made on image-formation hypotheses. If image color is not simply a result of color formation in the cellulose of the linen fibers, image formation must be a much more complex process than we originally thought.
Response
Here we see Rogers at his most infuriating – sounding so categorical, so insistent, so terribly-well informed – yet failing to explain, and using vague imprecise terminology.
OK, so there is banding, and that banding has been invoked to explain the cut-off nature of the face at each side which gives the face a less-than-real life appearance.
But how serious is it? Why has a uv illuminated picture been used to illustrate it. Uv and background fluorescence/quenching effects introduce all kinds of uncertainties. Would not a simple picture taken in reflected, scattered light have served better?
Go to Shroud Scope? Is there evidence there for serious banding effect, and if so, is it anything to do with yarn variation?
Another prime example of Rogers’ careless use of words, even chemical words. He refers to hemicelluloses as flax impurities – a flawed use upon which much extra confusion arises when he develops his “impurity layer” idea. Fact: hemicelluloses are NOT an impurity of linen. They are an intrinsic component. What’s more, they do not confer colour, so would not be a target for bleaching. I believe that Rogers was confusing or at any rate conflating hemicelluloses with pectins. The latter have some characteristics in common, notable pentose sugars, but their distinguishing characteristic is their acidic character and negatively charged groups. Their relevance to linen is not to do with colour. It’s to do with the fact that they are the matrix substance that hold the bast fibres of flax into bundles, and it is the purpose of retting to “rot” the wet flax stems so as to remove the pectins, separating the individual elementary flax fibres, which are then re-assembled in bundles by spinning to form threads but without that cementing pectin.
All of the bleaching processes used through history remove lignin and most associated flax impurities (e.g., flax wax and hemicelluloses). The more quantitative the bleaching process the whiter the product. The bands of different color on the Shroud are the end result of different amounts of impurities left from the bleaching process.
The bands of different color on the Shroud are the end result of different amounts of impurities left from the bleaching process.
Response: That may well be true. But beware a classic flaw in logic, which is to turn that statement around, and assume that any banding one sees on the Shroud must ipso facto represent batch-to-batch differences in the yarn. That is by no means certain or self-evident. For example, the cut-off effect on the two sides of the face is surely too bilaterally symmetrical to have arisen as the result of yarn differences.
Notwithstanding Anna Maria Donadoni , whose expertise I do not question, one also has to ask at what stage bleaching is likely to be carried out on linen, especially the higher grades (herring bone twill etc) – at the yarn stage – producing colour differences on the final cloth, or at the cloth stage to produce a homogeneous end result without a “quilt effect”. Surely the latter?
Where darker bands of yarn intersect image areas, the image is darker. Where lighter bands intersect an image area, the image appears lighter. This proves that the image color is not a result of reactions in the cellulose of the linen. Some impurities on the surface of the different batches of yarn produced the image color. This observation is extremely important when tests are being made on image-formation hypotheses. If image color is not simply a result of color formation in the cellulose of the linen fibers, image formation must be a much more complex process than we originally thought.
Here we see Rogers going into overdrive, and basing major conclusions on evidence that is at best impressionistic. Yes, one expects an additive effect of yarn and image colour. But we are then pitchforked into a conclusion of mind-boggling dogmatism, namely that “this proves that the image color is not result of reactions in the cellulose of the linen”. What possible justification can there be for such a conclusion – whether proved right or wrong in the fullness of time? What evidence is there so far that the colour is due to impurities on the surface as distinct from in the cellulose? None whatsoever that I can see. The “impurity layer” hypothesis of which so much use has been made, and which has been endlessly touted as if fact rests on some very dodgy, opaque, impenetrable exposition. Note: I am not disputing the position that the image is not in the core crystalline cellulose. But we moved on from that issue, and in any case there is cellulose in the PCW as well, albeit less crystalline. We seem to have another hotch potch of poorly presented, ambiguous misleading unstructured argument which is then used to underpin a highly individual and partisan position. That is why I have said so little re Rogers in the past – and the little I have said has usually been less than flattering. Regardless of the soundness or otherwise of his chemical know- how it is his flawed exposition that makes it so difficult to address the issue – not knowing what he really understood or misunderstood through his woolly imprecise use of words and syntax.
If image color is not simply a result of color formation in the cellulose of the linen fibers, image formation must be a much more complex process than we originally thought.
But that’s because Rogers showed no recognition of the PCW and its superficial reactive hemicelluloses. Had he recognized it, he might have seen image-imprinting as LESS rather than more complex, compared with imprinting onto a highly ordered crystalline cellulose. The reason it is “complex” is because Rogers has made a spectacular jump from intrinsic cellulose to extrinsic impurities, and from pyrolysis to Maillard chemistry requiring not only reducing sugars but putrefaction amines also. The latter “diffusion hypothesis” has since been elevated to holy writ and still dominates all the discussion on Shroud forums, despite being based on a faulty understanding of linen fibres and their microstructure.
Based on the facts of chemistry and current storage conditions, the Shroud of Turin is not now and has never been in imminent danger of catastrophic autocatalytic decomposition. The “restoration” of 2002 was based on an erroneous understanding of chemistry.
Autocatalytic chemical reactions are those in which the rate increases as the amounts of reactants decrease, i.e., while the materials are reacting. The most important single factor in predicting effects is the temperature. When the temperature changes, the rate changes. The only severe heating episode the Shroud has suffered was during the fire of 1532. Any autocatalytic decomposition that occurred then has long since stopped as the Shroud is stored at normal temperatures.
The fundamental chemical-rate equation that describes an autocatalytic process is the following:
where ? is the fraction reacted at any specific time, t. The derivative, d? ?dt, is the rate of the reaction. E is the “Arrhenius activation energy,” and Z is the “Arrhenius pre-exponential.” Each applies only to a single specific, consistent reaction being studied. The value of the “rate constant,” k is different at each specific temperature: It is a constant only at one temperature, and it applies only to one specific reaction. The values of E and Z are determined from a large number of k measurements at different temperatures.. Predictions of the Shroud’s expected lifetime can not be made on the basis of a single rate constant. Observations made during a scorching event can not be applied to rates at normal temperatures.
E, Z, and k are the most important values in a discussion of rates and associated lifetimes of materials. All of these values have fundamental meaning in the chemical reaction. R is the “gas constant (1.9872),” a universal constant that applies to many disparate physical and chemical processes, and it is known with great accuracy and precision. T is the absolute temperature, expressed in degrees Kelvin (0K = -273.2?C). The exponents p and q allow the prediction of the position of the maximum rate in an autocatalytic process, i.e., the amount reacted at the maximum rate – at constant temperature. Exponents higher than 2 are extremely rare.
Examples of simple and autocatalytic rate curves are shown in the figure. Notice that the rate increases with time in the autocatalytic curve, at constant temperature, until it reaches a maximum reaction rate. Then the rate decreases. However, the initial rate at any temperature is much lower than the maximum rate. The chemical decomposition rate of cellulose is essentially zero at room temperature. Most long-term degradation of cellulose that is observed in archaeological contexts is caused by microbiological attack.
When cellulose is decomposing autocatalytically at high temperature, the rate can be returned to its initial value by cooling.
Reaction rates in solids, especially crystalline solids like cellulose, are much lower than the values for the same material in a solution or melt, because a crystalline lattice is stabilized by its ordered structure. The crystal structure is called “fibrillar” in materials like linen.
A major cause for autocatalysis in cellulose decomposition is the destruction of crystalline order when the material is heated above its melting point, about 260ºC. With the exception of the fire of 1532, the Shroud has never faced this danger. Secondary, chemical autocatalysis is discussed below. Rates in the normal cellulose solid phase are essentially zero in the absence of acids, bases, short-wavelength light, or water and microorganisms.
When the crystalline order of cellulose is destroyed by heating, the cellulose melt is also chemically autocatalytic. The possibility for chemical autocatalysis in linen depends on the products of cellulose decomposition. Feigl and Anger [Feigl, F. and Anger, V., 1966, Spot Tests in Organic Analysis, Elsevier Pub. Co., New York.] describe the effects of heating cellulose as follows: “When cellulose is heated it decomposes and the resulting superheated steam reacts with unchanged cellulose to produce hexoses, which in turn hydrolyze to give hydroxymethylfurfural.” The only important chemical catalyst for the autocatalytic degradation of cellulose at high temperatures is superheated steam. Superheated steam does not exist at room temperature. There is no “memory effect.” The Shroud should be as stable at room temperature as any other sample of linen. The Shroud was in no danger of autocatalytic decomposition.
The decomposition rate of a crystalline solid depends on crystal perfection. When crystals are put under stress, they develop high-free-energy defects, and decomposition is much faster at the defects than it is in the parent material. If autocatalysis were a real problem for the Shroud, significant differences should have been observed around the stressed and strained stitching of the patches. STURP observed those areas, and there was no sign of accelerated autocatalysis, indeed there is no sign of any autocatalysis. Autocatalysis is not a real hazard for the Shroud.
More detailed studies have shown that the major or secondary products of the thermal decomposition of cellulose are formaldehyde, furfural (2-furaldehyde), hydroxymethylfurfural (5-hydroxymethyl-2-furaldehyde), levulinic acid (4-oxopentanoic acid), and 3-pentenoic-?-anhydride. None of these are a significant catalyst for the autocatalytic decomposition of linen. Indeed, formaldehyde, furfural, and hydroxymethylfurfural are reducing agents, antioxidants. Furfural inhibits the growth of molds and yeasts. Scorched areas are less likely to show microbiological attack.
Observations and descriptions of the Shroud through the 470 years since the fire of 1532 do not support fear of catastrophic decomposition of the cloth. There is absolutely no evidence for attack on the cloth by acids, bases, or microorganisms. Samples from all parts of the Shroud were tested for pH by STURP. No impurities that could start autocatalytic decomposition were found, confirming what was observed through the 470 years of history.
If Shroud deterioration is still a worry, one practical way to slow the rate is to keep it cold. That also has the advantage of reducing microbiological attack. As in the case of the use of “inert” atmospheres, storage at reduced temperature should carefully be considered. Too low a temperature could cause physical stress and might cause fibers to fracture. It would probably cause the thin coating of image color on the fibers to be loosened in some areas.
As a rule of thumb according to the Arrhenius expression, rates of normal reactions are increased by a factor between two and three for each 10?C increase in temperature. Some moderate cooling could have a significant effect on prolonging the life of the Shroud. Severe freezing could damage the cloth and image.
Latest addition: 16:30
My reply: Important if one is a Shroud custodian, charged with keeping this fascinating artefact as intact as possible for future study (shame that a lot of present data is being withheld).
Won’t address the complex issues involved point-by-point, though may return on a future occasion. Simply say this for now:
Why no mention of light, especially sunlight, especially uv. No mention of lignin either– well known in the context of yellowing of paper. Odd, because of Roger’s interest in vanillin – a breakdown product of lignin. Protective argon helps – baecause uv sensitization is only the first step in a process that may have several steps, one of more involving oxygen. But an alternative to oxidation is free-radical mediated chemical cross-linking, so prime consideration is to protect the Shroud from light.
The argon not only protects from oxygen. It protects from atmospheric pollutants, like ground-level ozone (that adds across double bonds forming ozonides), nitrogen dioxide and sulpur dioxide (forming oxy-acids in the presence of moist air). Microorganisms (fungi etc) ? The decision to use thymol highly regrettable. Should have been non-organic, i.e. non-carbon based to avoid contaminating with still more modern carbon that might compromise a new round of radiocarbon dating.
 Latest addition: 16:00 5th Jan
Latest addition: 16:00 5th Jan
The 1988 radiocarbon age determinations were carefully done. The sample preparation methods, the measurement technologies and procedures, and the data reduction were adequately planned and executed to answer the most important question: was the Shroud produced in the First Century? Damon, et al., reported that “The age of the shroud is obtained as AD 1260-1390, with at least 95% confidence.” However, that date does not reflect observations on the linen production technology nor the chemistry of fibers obtained directly from the main part of the shroud in 1978. The independent analyses from the different laboratories scatter more than would be expected for a homogeneous sample, raising other questions.
The 1988 sampling operation was described as follows: “The shroud was separated from the backing cloth along its bottom left-hand edge and a strip (~10 mm x 70 mm) was cut from just above the place where a sample was previously removed in 1973 for examination. The strip came from a single site on the main body of the shroud away from any patches or charred areas.” The use of a single sample, assuming it was representative of the whole cloth, defied normal procedures and protocols established before the radiocarbon study. It was a serious mistake.
To make matters worse, Mssrs. Franco Testore, professor of textile technology at the Turin Polytechnic, and Gabriel Vial, curator of the Ancient Textile Museum, Lyon, France, approved the location of the radiocarbon sample without any serious attempt at characterizing the sample. No chemical or careful microscopic sample characterizations were made. The 1988 work did not guarantee the validity of the sample.
The area where the radiocarbon sample was obtained had been photographed in 1978 with an ultraviolet source (see “UV fluorescence”). While making the UV photographs, the source was heavily filtered to exclude visible light and the camera was heavily filtered to exclude any effect of the UV on the film. All that appears on the film is the result of pure fluorescence. All fluorescence is a result of the chemical composition of the material.
The non-image cloth typically shows weak fluorescence (upper right). When image appears on the cloth, it quenches the fluorescence and gives it a brown color (see “Hands” below). The small, triangular, white area is where the Raes sample was cut in 1973. The radiocarbon sample was cut upward from there about 1 cm to the right of the seam and about 7 cm long. The area where the radiocarbon sample was taken is relatively dark, a fact that is not the result of dirt, image color, or scorching. The cloth is much less fluorescent in that area, brightening into more typical fluorescence to the right. The photograph proves that the radiocarbon area has a different chemical composition than the main part of the cloth. This was obviously not considered before the sample was cut.
Raes and radiocarbon yarn show colored encrustations on their surfaces. Some sections of medulla contain some of the material, showing that it had been able to flow by capillary attraction as a liquid. The encrustation is not removed by nonpolar solvents, but it swells and dissolves in water. There was absolutely no encrustation on either the Holland cloth or fibers from the main part of the Shroud.
Al Adler had found large amounts of aluminum in yarn segments from the radiocarbon sample, up to 2%, by energy-dispersive x-ray analysis. I found that the radiocarbon sample was uniquely coated with a plant gum (probably gum Arabic), a hydrous aluminum oxide mordant (the aluminum found by Adler), and Madder root dye (alizarin and purpurin). Nothing similar exists on any other part of the Shroud. The photomicrograph shows several fibers from the center of the radiocarbon sample in water. The gum is swelling and slowly detaching from the fibers. Many red alizarin/mordant lakes can be seen, and yellow dye is in solution in the gum. Several cotton fibers are visible, a situation unique to the Raes and radiocarbon samples.
The radiocarbon sampling area had been dyed to match the old part of the cloth. The sample chosen for dating was totally invalid for determining the true age of the Shroud.
My answer, now as always: if there are any doubts as to the representativeness of the sample taken (as indeed there should be, given the extraordinary decision to restrict sampling to one corner) then repeat the entire procedure. One could take multiple samples from the ‘less iconic’ dorsal side. One could use cellulose-solubilising reagents, e.g. cuprammonium hydroxide, to improve the efficiency of the clean-up procedures.
As discussed in (3) above, the crystallinity of the flax fibers in all of the parts of the Shroud that were not scorched has not been significantly degraded.
My reply: added at 12:00:
Again, hardly a user-friendly introduction. One has first to realize that Rogers is referring (apparently) to the 1532 fire in that reference to parts of the Shroud that were NOT scorched – hardly helpful if addressing the hypothesis that the image itself is a scorch, albeit of much earlier provenance. Crystallinity of flax fibres presumably means cellulosic microfibrils of the secondary cell wall of flax fibres – but these are in the core of fibres. What about the superficial parts of fibres that are or should be the major focus if one is attempting to explain how fibres acquired their yellow or brown coloration?
“Crystallinity… not significantly degraded”? Ouch. Does he mean “little or no loss of native crystallinity”. Those of us who know a little about crystallinity in carbohydrates learn to be cautious in our use of words, given that a crystal may temporarily “melt” and subsequently recrystallize to a new ordered state that looks at first sight like the original crystal form but may be subtly different. (See the extensive literature on starch retrogradation for which there are some parallels with more ordered cellulose).
The Arrhenius Law describes the effect of temperature on rate constants for all consistent chemical reactions, as follows:
k = Ze-E/RT
where k is the rate constant at any specific temperature, Z is the Arrhenius pre-exponential (related to the probability that any specific molecule(s) will react), E is the Arrhenius activation energy, R is the gas constant, and T is any specific, constant absolute temperature (degrees Kelvin).
My reply : 12:08
Where is this leading? Pretty well every chemical reaction involving collision between molecules has an Arrhenius energy “hump” aka ‘energy of activation’ that has to be overcome, expressed mathematically by the Arrhenius equation.
If the image were a scorch or any part of the Shroud had been heated enough to make significant changes in the rates of decomposition of any of its components, we would see changes in the structure of the flax fibers and blood.
Wrong! Totally and utterly wrong! That a thermochemist should write this is astonishing. The temperature term in the Arrhenius equation is part of an exponential – so small differences in temperature produce LARGE changes in rate of reaction. As a rule of thumb, a 10 degree rise in temperature typically doubles or even trebles a rate of reaction. The pyrolysis temperature of hemicelluloses is reported as 220 degrees. That of cellulose as 280 degrees. That’s a 60 degree difference in temperature: 10: double, 20: fourfold, 30: eight fold, 40: 16 fold, 50: 32 fold, 60: 64 fold. So by temperature control it is possible to get selective pyrolysis of hemicelluloses with scarcely any pyrolysis of cellulose.
Hemicellulose degrades easily when heated, far more so than cellulose, and at a lower temperature. The respective temperatures for the onset of pyrolysis (“scorching”) measured as weight loss are 220-315C for hemicellulose and 315-400C for cellulose respectively (H.Yang et al, 2007).
My reply: added at 12:10
Let’s take the average value for hemicelluloses, i.e. 268 and the lowest value for cellulose i.e. 315. That’s an approx 47 degree difference – with an expected 30 fold difference in rate of reaction.
Methinks Mr.Rogers must have been having an off-day when he wrote his lecture notes on the Arrhenius equation – having brought in theory with a view to dismissing scorching when- if anything – it should have flagged up the non-cellulosic polysaccharides as a prime target for selective pyrolysis, especially if they have a superficial location (which indeed they do, though that leads on to another major blind spot on Rogers’ part).
The blood still evolves hydroxyproline on mild heating, and the cellulose crystals are largely undistorted. Image and control fibers show identical crystal properties. The image is not a scorch. The cloth was not heated, not even boiled in oil.
Where does one begin? Firstly, hats off to Rogers for detecting hydoxyproline (HP) in a Shroud ‘bloodstain’– more correctly a fragment in pyrolysis-mass spectrometry that could be matched with nothing else of likely physiological relevance. It could provide a crucial clue to the nature and origin of the Shroud ‘blood’. But one thing at a time we’ll return to HP later. First, it has to be said that Rogers made a MAJOR error. He assumed it HP was a normal constituent of blood. What’s more, he must have picked up on the fact that the meat industry uses HP as a marker for HEATED as distinct from uncooked meat. But HP is NOT a marker for blood – not even the blood that is inside meat, ie. muscle/connective tissue. (whose pink or red colour is incidentally due as much to muscle myoglobin as entrapped blood haemoglobin). You see, HP is a marker for connective tissue – especially collagen, of which there is plenty in meat but not blood per se. As I say, we can discuss another time what HP was doing in Shroud blood –I have produced a novel off-the-wall explanation elsewhere.
Suffice it to say that no sooner had Rogers stumbled on HP in Shroud “blood” than he was pressing it into action as an argument for Shroud blood never having been exposed to heat. Not only were his assumptions/first impressions wrong – HP is not a normal component of blood except in trace amounts – and is difficulty volatile (MPt 275C with decomposition), he was impetuous and some might say cavalier in attempting to use it as an assay procedure – one that had never been properly evaluated and accredited. (He later did the same with his vanillin). The term “loose cannon” is one that springs to mind. Or should that be chemical prima donna?
“Cellulose crystals largely undistorted”. We have dealt with that already. It’s the hemicelluloses that should be the prime concern. Rogers himself said as much later – see above, ed. No. x. (insert later)
“The image is not a scorch etc” . The scorch hypothesis has been dismissed with indecent haste, invoking largely irrelevant – some might say spurious evidence. The bit about “boiling” is presumably a reference to the Savoy courtier who in 1503 enthused about the “trials” inflicted on the Shroud, some 150 years after its first recorded display at Lirey in 1355 approx. The possibility that the Shroud was once boiled in oil is intriguing – something to be considered carefully given its ability to alter the Shroud image – but ought not not casually slipped into a supposedly scientific argument for dismissing the scorch hypothesis.
 The primary effect of all kinds of radiation is to heat the material it hits. This statement includes electromagnetic radiation (visible, ultraviolet, and infrared radiation); ionizing particles such as protons, electrons, and alpha particles; and non-ionizing particles such as neutrons. You can feel the heat when you hold a lump of plutonium, a flask of tritium, or a recently irradiated accelerator target. Intense irradiation can cause enough heat to explode explosives and burn metals (think of laser effects).
The primary effect of all kinds of radiation is to heat the material it hits. This statement includes electromagnetic radiation (visible, ultraviolet, and infrared radiation); ionizing particles such as protons, electrons, and alpha particles; and non-ionizing particles such as neutrons. You can feel the heat when you hold a lump of plutonium, a flask of tritium, or a recently irradiated accelerator target. Intense irradiation can cause enough heat to explode explosives and burn metals (think of laser effects).
My reply – added at 11:30
True as far as it goes, but an awful lot of detail is omitted, especially as regards the more energetic short wavelength/high frequency radiation which tends to produce free radicals by breaking chemical bonds. What’s more, there needs to be a light-absorbing chromophore present for each class of radiation that can absorb that radiation, an essential preliminary to producing heat. Some substances are essentially transparent to uv radiation, say, lacking as they do an appropriate chromophore or having exceptionally strong chemical bonds with large dissociation energies.
White linen is largely reflective. There is little heating effect when ordinary visible white light of infrared rays fall on it, since most is scattered/reflected. Of course, you can heat it up by using intense sources, based on the fact that a mere 1% absorption, say, will have a heating effect if it is 1% of a large number. (In passing: my own experiments with radiation – from an infrared/visible emitting spotlight- used charcoal to get local scorching. Without the charcoal to absorb the radiation there was no visible scorching).
Cellulose molecules are folded back and forth in a fairly regular arrangement, and they show the properties of crystallinity. This is called a “fibrillar structure.” When you rotate the stage of a petrographic microscope with crossed polarizers while looking at a linen fiber, straight lengths change from black through colored to black again every 90?. The fiber is birefringent and has an ordered structure.
When cellulose fibers are heated enough to color them, whether by conduction, convection, or radiation of any kind, water is eliminated from the structure (the cellulose is “dehydrated”). When water is eliminated, C-OH chemical bonds are broken. The C? free (sic) radicals formed are extremely reactive, and they will combine with any material in their vicinity.
In cellulose, other parts of the cellulose chains may be the closest reactants. The chains crosslink. Crosslinking changes the crystal structure of the cellulose, and you can see the effect with a polarizing microscope.
When cellulose starts to scorch (dehydrate and crosslink), its characteristic crystal structure becomes progressively more chaotic. Its birefringence changes, and not all parts of a straight fiber go through clear transitions from dark to light at the same angle. Zones of order get smaller and smaller. It finally takes on the appearance of a pseudomorph and just scatters light.
My reply: added at 11:35
Yes, but it requires high temperatures or other energetic sources to degrade cellulose. So why focus on cellulose at this stage when there are other more reactive constituents in linen, such as the non-cellulosic carbohydrates (pectins, hemicelluloses etc) and lignin (an aromatic non-carbohydrate). Yes, this is acknowledged later, but is an instance of Rogers’ somewhat unhelpful style of writing that lurches between this kind of needless padding and then cursory references to the matters of real interest and concern. More later:
A significantly scorched fiber does not change color as the stage is rotated between crossed polarizers. Specific types of radiation cause specific types of defects in the crystals of flax fibers. For example, protons ionize the cellulose as they pass through the fiber. This warps the crystals, making the protons’ paths birefringent. You can see where they went in the fiber by the straight lines of their paths (see the “Proton-irradiated” figure in source material).
Not all kinds of radiation ionize the material they penetrate. Neutrons and neutrinos do not have any electrical charge. Neutrinos hardly interact with matter at all, the fact that made them so difficult to detect. They have practically no chance of being stopped as they shoot through the entire diameter of the Earth. The effects of neutrons depend on their energy, but they normally interact with hydrogen-containing materials to produce “recoil protons.” They knock a hydrogen nucleus out of the material, producing an ionizing proton. You can see the ionization streaks of these (usually lower energy) protons (see the “Neutron-irradiation” figure see original source ).
My reply: added at 11:37
Again, all of questionable or dubious relevance to the Shroud image – especially as it “does not lead anywhere”. More to follow later
The crystal structure of the flax fibers of the Shroud shows the effects of aging, but it has never been heated enough to change the structure. It has never suffered chemically significant irradiation with either protons or neutrons. No type of radiation that could produce either color in the linen fibers or change the 14C content (radiocarbon age) could go unnoticed. All radiation has some kind of an effect on organic materials.
My reply: added at 11:45
Confusing, and where the last statement is concerned – somewhat self-contradictory and incorrect. All high-energy radiation has some kind of effect maybe, but visible light and infrared etc usually have negligible effect unless exceedingly intense or one is talking about centuries of exposure. Yes, the Shroud is centuries old, but why are we being burdened with largely conjectural age-related effects of questionable relevance when the key issue is one how the image was formed. This is yet another instance of Rogers’ irritating and bewildering now-you-see-it-now-you-don’t style of exposition – his failure to present cogent, focused and, most of all – structured arguments. More later.
This proves that the image color could not have been produced by thermal or radiation-induced dehydration of the cellulose. Image formation proceeded at normal temperatures in the absence of energetic radiation of any kind.
My reply: added at 11:50
Finally we get to the nitty gritty – but find a statement that is virtually impenetrable. Nothing can be “proved” or disproved at this stage, given that there has been only the most superficial analysis of what may or may not have caused the coloration of Shroud fibres. “Thermal or radiation-induced” dehydration of cellulose? But thermal energy IS radiation if projected across an air gap, and given there’s no mention so far of conduction by direct contact (my preferred hypothesis) why try to make “thermal” energy sound different from light or uv – given it’s all part of the same continuous electromagnetic spectrum? And why this focus, fixation some might say, on the dehydration of cellulose? Why dehydration – given that we have been told nothing substantive so far about the nature of the coloured image? Why the focus (still) on cellulose, given its just one carbohydrate and the least likely to be altered by exposure to heat, or high-energy radiation?
More to follow
Added at 11:00 5th Jan (rest to follow later)
Alan Adler was an expert on porphyrins, the types of colored compounds seen in blood, chlorophyll, and many other natural products. He and Dr. John Heller, MD, studied the blood flecks on the STURP sampling tapes [Heller and Adler, Applied Optics 19, (16) 1980]. They converted the heme into its parent porphyrin, and they interpreted the spectra taken of blood spots by Gilbert and Gilbert. They concluded that the blood flecks are real blood. In addition to that, the x-ray-fluorescence spectra taken by STURP showed excess iron in blood areas, as
expected for blood. Microchemical tests for proteins were positive in blood areas but not in any other parts of the Shroud.
Several claims have been made that the blood has been found to be type AB, and claims have been made about DNA testing. We sent blood flecks to the laboratory devoted to the study of ancient blood at the State University of New York. None of these claims could be confirmed.
The blood appears to be so old that the DNA is badly fragmented. Dr. Andrew Merriwether at SUNY has said that “… anyone can walk in off the street and amplify DNA from anything. The hard part is not to amplify what you don’t want and only amplify what you want (endogenous DNA vs contamination).” It is doubtful that good DNA analyses can be obtained from the Shroud.
It is almost certain that the blood spots are blood, but no definitive statements can be made about its nature or provenience, i.e., whether it is male and from the Near East.
My answer to FAQ 2:
“They concluded that the blood flecks are real blood.” That statement is potentially misleading and goes beyond the data. For a start the summary omits all mention of the unexpected findings – atypical porphyrin spectrum, absence of potassium, failure to see any cells etc. It mentions only the few positive findings. Accompanied by a hypothesis that places a central role for bilirubin – subsequently promoted as if experimental fact – despite there being no quantitative measure of bilirubin, and indeed the evidence for any bilirubin at all appears to rest on dubious uv fluorescence characteristics and a spot diazo test (not reliable except on authenticated blood).
I have previously described the bilirubin story as pseudoscience. Unflattering, but still my position. It is the pseudoscience that first got me interested (again) in the TS which I consider that scientists have a duty to expose (especially retired ones like myself who need have no fear of damaging their credibility in their own specialized field through being seen as “controversial” or “publicity-seeking”).
I believe that what is one the Shroud is somewhat altered blood, but was probably not blood directly from a living or recently deceased subject. I shall leave at that – not writing this to promote my own theories.
(posted at 09:55 Sat 5th Jan)
The primary goal of STURP was to test the hypothesis that the Shroud’s image was painted, as claimed by Bishop d’Arcis in 1389. If it had been painted, some colored material had to be added to the cloth, but the colored material would have gone through the fire of 1532. The pigments and vehicles would have suffered changes in response to the heating, the pyrolysis products, and the water used to put the fire out. No changes in image color could be observed at scorch margins. We tested all pigments and media that were known to have been used before 1532 by heating them on linen up to the temperature of char formation. All of the materials were changed by heat and/or the chemically reducing and reactive pyrolysis products. Some Medieval painting materials become water soluble, and they would have moved with the water that diffused through parts of the cloth as the fire was being extinguished. Observations of the Shroud in 1978 showed that nothing in the image moved with the water.
The Shroud was observed by visible and ultraviolet spectrometry, infrared spectrometry, x-ray fluorescence spectrometry, and thermography. Later observations were made by pyrolysis-mass-spectrometry, lasermicroprobe Raman analyses, and microchemical testing. No evidence for pigments or media was found. Your eye sees colors when the surface absorbs some wavelengths of light and reflects others. A red surface absorbs all visible wavelengths other than red. Each chemical compound absorbs wavelengths that are characteristic of its chemical structure. The best way to determine the properties of a color is by measuring its spectrum. Reflectance spectrometry was one of the most important contributions of the STURP observations. The reflectance spectra in the visible range for the image, blood, and hematite are shown in the figure. The image could not have been painted with hematite or any of the other known pigments. The spectrum of the image color does not show any specific features: it gradually changes through the spectrum. This proves that it is composed of many different light-absorbing chemical structures. It has the properties of a dehydrated carbohydrate. (ed. my emphasis)
There is no evidence for significant amounts of any of the many pigments and/or dyes that could have been used to paint or touch up the blood stains. We had considered and studied Tyrian purple (6,6′-dibromoindigo) and Madder root dye on an aluminum and/or chromium mordant as well as cinnabar (mercuric sulfide) and ferric oxide pigments. During and before the 14th Century, gold metal was the most important yellow. That would easily be detected by x-ray fluorescence. Other pigments in common use were yellow ocher (hydrated Fe2O3), burnt ocher (hematite Fe2O3) and other ochers, orpiment (As2S3), realgar (AsS), Naples Yellow (Pb3[SbO4]), massicot (PbO), and mosaic gold (SnS2). Organic dyes included saffron, bile yellow, buckthorn, and weld. Madder root began appearing in Europe from the Near East about that time. Many of the dyes required mordants, which are hydrated oxides of several metals (e.g., aluminum, iron, and chromium). In order to produce the shadings observed in the Shroud’s image, the concentrations of pigments would have to vary across the image. No variations in any pigment were observed by x-ray fluorescence spectrometry. The image was not painted with any inorganic pigment of an appropriate color.
My (preliminary) response:
Given the amount of experimental evidence that Rogers marshals against an artist’s pigment, none of which incidentally I dispute, is it not rather odd that Rogers slips in the bald statement “It has the properties of a dehydrated carbohydrate” (emphasized in bold above) withouth further explanation?
Speaking as someone who has argued that the Shroud image is a scorch – peremptorily dismissed by Rogers – I too believe, or rather ASSUME that the image to be dehydrated carbohydrate – though with precious little by way of dierect evidence – except that which derives from model systems with modern linen. The evidence that the Shroud image is a scorch, representing (chemically) dehydrated carbohydrate is arrived at mainly by a process of elimination (admittedly less than ideal, and indeed a source of frustration). So what are the “properties” to which Rogers refers? Why is there no hint of what he means by “dehydration”, especially as he later goes on to dismiss “simple” pyrolysis in favour of a Maillard reaction, despite both involving “dehydration” steps*. (That’s “simple” in the sense that pyrolysis – thermal degradation – will always occur if the temperaure is high enough, regardless of whether other substances – notably oxygen – are present or absent, whereas a Maillard reaction requires more than raised temperature – needing a source of reducing sugar and an amine – both of which are highly conjectural where the Shroud is concerned)
I would have preferred a less wordy introduction, and not having to deal immediately with the minutiae of chemical reactions – this posting being focused mainly on botany and cell walls – but that’s for the reason stated – I am following Rogers’ script in his order).
This failure to talk around crucial aspects, to introduce them suddenly without preamble -and then immediately move on, is a recurring feature in his writing – as will be seen – and I have to say not a welcome one.
* I suspect that his case for “dehydration” would have rested on Adler’s discovery that image coloration is bleached by diimide, HN=NH. That involves a sequence of assumed chemical reactions, i.e. in which unsaturated (hydrogen-deficient)double bonds are introduced by loss of the elements of water – 2H per O atom – creating a coloured chromophore, with diimide reversing the process by adding back hydrogen across the double bonds. Whilst I would not have expected Rogers to add anything like that degree of detail, I do believe he should signalled that the evidence for chemical dehydration was suggestive, or even highly suggestive, rather than proven. Or maybe I’m old-fashioned in believing in a strict demarcation between hypothesis and fact – which goes some of the way to explaining why I am so alarmed at the extent to which so many otherwise thoughful and level-headed folk appear to have gone lock, stock and barrel for Rogers’ (highly tendentious) ideas…
























Golly. Well, a bit at a time…
That UV picture… it’s glowing so EXACTLY like a scorch…
I now have a coloured copy of Pellicori and Miller’s paper, and although I agree completely with their observations, I disagree almost as completely with their conclusions.
Surely it’s the background only that glows, Hugh, with that fluorescence being quenched by the body image? In other words, exactly as stated by the critics of scorching . Sigh…
PS: I’m not enjoying the present task one little bit – but someone has to prick that balloon…
Pellicori and Miller do indeed say that the background glows and image does not, which is true. However, to conclude that this observation precludes a scorch is, I think incorrect. My scorching experiments on linen invariably result in something very similar – a dark scorch surrounded by “background” fluorescence. On the shroud, this background fluorescence extends well beyond the image, so was probably produced separately, but not necessarily by a different mechanism. Pellicori and Miller suggest that it is due to aging, but admit that exactly the same effect can be achieved using heat. What’s more, their photographs show the background fluorescence decreasing at the edges of their photos, and especially at the corners of the cloth, which may suggest that it is not as uniform as would be expected if the whole cloth had been similarly treated. (It may also suggest that the UV light was spotlighted in the middle and didn’t reach the edges, but they don’t mention that possibility. There is a whole subsection of the literature about what this ‘edge-decrease’ might mean regarding the C14 sample, but it might not really be there at all!)
They also observe that the big scorches on the shroud (but not the minor ones) actually fluoresce themselves, and there is a massively enhanced photo (ohioshroudconference.com/papers/p07.pdf) which does show red fluorescence from the darkest parts of the 1532 burn marks. The rest of the discolouration, image, blood or scorch, does not fluoresce, although there is a bright halo around the outer edge of the spear-wound. I still find this red fluorescence very odd, and wonder if it is not simply the reflection of the red visible light shining off the wholly (shiny?) carbonised areas.
If what you say is true, Hugh, then Barrie Schwortz is going to have to update or even rewrite his script.
Sorry for the short reply. There’s much to reflect on in your comment which I may comment on later. But first I must bite the bullet again today, and try to complete my critique of Ray Rogers’ FAQs.
I think there is a tendency to misremember (or even misread) papers that came out a long time ago, and they only need to be quoted once or twice for the misinformation to be set in stone. Barrie Schwortz’s comment-8125 is a case in point.
– “true scorches on linen will always fluoresce in the red.” This is obviously nonsense, and even Pellicori and Miller don’t claim that it is true.
– “Every documented scorch on the Shroud fluoresced in the red.” As above.
– “The only conclusion possible from these observations is that the Shroud image is NOT the product of scorched or heated linen.” The observations being false, the conclusions are wholly unjustified.
May I quote you (oh kindred spirit)? Yours is what the Readers Digest used to call a “quotable quote”.
Feel free. The evidence is available. Pellicori and Miller distinguish very clearly between the 1532 scorches (which fluoresce red – but check out the enhanced photo from ohioshroudconference) and the Lirey poker holes, which don’t. They attribute this to the Lirey holes being more burnt than scorched. However they also find a couple of ‘donut-shaped’ areas which ‘appear to be scorches’ although they are ‘brightly fluorescing yellow-green.’ In laboratory tests, they found that “scorches emit a bright greenish-yellow fluorescence if produced in air and reddish if produced under conditions of limited available oxygen.” It is not clear to me if this means that they found that the brown scorches in visible light (darker than the background) were yellow in UV, lighter than the background. The only time I have achieved this is with cotton, not linen.
Dear Hugh,
Why is the question of the background fluorescence so important ?
What is you hypothesis to explain it ?
Best regards.
Thibault.
I don’t think I have a working hypothesis at the moment, but feel that the rejection of non-working hypotheses might help, whether they are mine or someone else’s. The importance of it is simply that it occurs at all. Is fluorescence a feature of all old linen? Has anybody asked a museum? If not, then how did it occur? I thought it might be in association with the body image, but it seems to be spread too widely over the cloth. Then I thought it might have been due some anointment, as the M&P images seemed to show it concentrated in the middle of the cloth – however, as I now see that the top, bottom and left edges of every photo are dark (not just the famous corner where the C14 came from) I think the fluorescence may cover the whole cloth after all, and the fringe darkness is an artefact of the photography. So is it a common feature of 1st century middle eastern or 14th century French cloth? Has anybody checked? If not, then the unusual preparation of the cloth may be significant. M&P say that they can replicate the background fluorescence with heat (which they don’t believe happened), but they do not demonstrate that it can be achieved by ‘aging’ (whatever that means). Since the shroud has spent most of its life rolled up, I would expect some sort of banding, darker on the outside and lighter in the middle, if light, heat, oxygen or other gas were responsible for ‘aging’ the shroud over hundreds of years. If it was used as the famous mandylion, then there should be a stripe across the face, if as a Byzantine altarcloth (folded in four as per Lirey holes) then one quarter might look more ‘aged’ than the other three, and so on. None of this seems to be the case.
Pingback: Colin Berry Takes On Banding and More « Shroud of Turin Blog
Hello Colin, here’s a comment on FAQ 17 point 3 on electromagnetic radiation vs corona discharges:
In an electrostatic field the locations of preferred discharge are the most protruding and pointed places: a lightning bolt will rather strike a bell tower than a house standing beside it. “A discharge may exist in the vicinity of a sharp point, or other place with a small radius of curvature where the electric field is increased significantly from its average value” (Dr. Calvert on electrical discharges). The electrostatic field lines are not straight but curved lines that bend to the most protruding, most pointed points, also if these are threads or even fibers that are just slightly more protruding than others (see again fig. 5 p.7 of CD-paper). The geometry of the cloth surface determines the distribution of the “surface electrostatic stress (surface electric field intensity)” (CD-paper p. 4, points 3, 4 and 6) and therefore determines the distribution of the filamentary streamers of electric charge toward the cloth. Electrostatic field lines and discharge streamers are not the same as electromagnetic radiation that ‘fires’ along straight lines. Discharge streamers don’t penetrate the cloth either. In the CD model and experiments “The glow-CD layer is interrupted by the presence of the TS and re-starts on the outer surface of the TS” (CD-paper p. 9). Any discharge that hit the reverse of the Shroud would have come from the reverse side, from the outside of the doubled Shroud.
In a corona discharge, the accompanying UV luminescence is generated inside the streamer, also where it hits the cloth: it would not radiate from the body itself. A CD is also accompanied by heat and “aggressive chemical by-products” (CD-paper p. 18-19).
That must rate at least 9/10 on the David Icke scale of wackiness, Adrie. I’m not saying it’s totally impossible. It’s not impossible that all the molecules in my house simultaneously deflect off their neighbours in the direction of open doors and windows, leaving me to inflate and perhaps explode in the vacuum thus produced, but sadly for some of my detractors the chances of that happening are 1 in a gadzillion, the latter further raised to the power of a squillion (but not impossible).
Professor Fanti is not a scientist – that much is clear from reading his papers full of unsupported claims. He’s a mechanical engineer who frankly hasn’t the first clue about the handling of data and other evidence or of the scientific method. If he’s so certain about corona discharges, then why is he not pressing the Shroud’s custodians to get the radiocarbon dating repeated. Once he gets his confirmation that the Shroud really is 2000 or so years old, then mainstream science bods like myself might be forced to give a second or two for considering some of the off-the-wall claims, corona discharges, seismic activity, radon etc etc.
I’m still in the process of tidying up my responses to those FAQs, Adrie, and will probably repost in standard format.
As a response I invite you to take a look at this photo of a corona discharge or at fig. 3 of the CD-paper (p. 5). They show deflecting streamers. The streamers are initiated by electrons, hitting air molecules/atoms, knocking out a free electron and leaving a positively charged ion behind. The positive ion and the negative electrons will subsequently preferrably move along the electric field line at the point of collision, but in opposite directions. When somewhere on its pathway one of the new free and electrostatically accelerated electrons collides with another air molecule, this molecule will in its turn ionise and produce a new free electron and ion, which start a new path way, in the direction of the field line at the point of this second collision. Then, somewhere on this new pathway a next collision will occur to the next air molecule, and so on. The glow of the corona is caused by electrons recombining with positive ions to form neutral atoms, which are not accelerated by the electric field (Mechanism of corona discharge).
The streamer is not a long continuous flow of molecules, but a chain of short pathways of separating and recombining ions and electrons along the local electric field line. The air pressure effects are minimal and wavelike: a corona discharge produces accoustic emissions.
Your responses to Rogers’ FAQ-paper are thorough and in a way necessary, Colin. Nevertheless, I have a few more points I have to address, among which one about first century Jewish provenance.
I’m sorry, it’s fig. 3 of Fanti’s second CD-paper of 2008.
I think there’s been a tiny misunderstanding, Adrie. It’s not the existence of corona discharges I question. It’s the claim that they created an image some 2000 years ago on the length of linen currently residing in Turin Cathedral.
The TS image has at least some of the major characteristics of a thermal imprint, as though produced by direct contact with a hot solid template. A thermal imprint off a bas relief template could account for the negative light/dark reversal. As for the ‘encoded’ 3D, that could be nothing more than applying software that converts image density to height on a 3rd dimension. – hardly rocket science (even if was NASA that supplied the original software!).
Granted, there are some unexplained differences re fluorescence or lack thereof between the Shroud image and its own scorch marks from that fire, the so-called “poker holes” etc (1532 provenance and earlier). But the latter were not scorches produced under known laboratory conditions, and in any case the precise chemical nature of the fluorescent species on the Shroud is unknown.
The extreme superficiality of the Shroud image (200-600nm?) is interesting, but I have little doubt that some of the faint scorches that one can produce on linen, assuming one’s parents did not christen one Paolo Di Lazzaro, are also superficial (where’s the evidence to the contrary, not counting ENSA scientists working on the TS as a side-line?).
Feel free to raise extra points re Rogers FAQ. I’m curious to know what new evidence you can bring to bear on the question of its age – assuming it’s not purely negative, e.g. finding still more fault with the radiocarbon dating. Why has that not been repeated? Message to Shroud custodians and the 3 laboratories (Arizona/Oxford/Zurich):
If a task is once begun
Never leave it till it’s done
Be the labour great or small
Do it well or not at all.
Hello Colin,
Would you please be so kind again to paste another e-mailed comment here? Thanks.
Reply: with pleasure Adrie. Here it is (I’ll read and, if need be, comment later):
Here’s a comment to FAQ 15 on first-century features, and FAQ 13 on starch evidence:
STARCH
Both Kohlbeck and Rogers found starch in an image area and a non-image area. They both got a positive standard starch test (deep red) from non-image fibers. These fibers were from 14 Raes threads – not C14 threads –, which Rogers got from Gonella in 1979 (A Chemist’s Perspective, p. 64), and which I assume authentic and not part of a repair). Kohlbeck’s photo slide #42 shows that all ca. 15 fibers in view are completely red-brown; better visible on the third slide on this HSG-page (HSG password request form). Also McCrone and Adler independently reported the presence of starch on the Shroud. For details see Internal Selvedge, 2.2 and 4.2.1. Rogers found that the starch was insoluble in concentrated HCl = hydrochloric acid and that the coating completely surrounded all surface fibers of his Raes sample and even was sometimes inside the fiber, as if wiped on as a viscous paste (Scientific Method, p. 17-20, cf. my comment of Jan 3 and the SEM fiber cross sections in Brown, Microscopical Investigation, p. 7-8, and, if you like, Baldan’s SEM photos of Shroud fibers furnished by Riggi di Numana on Fanti’s homepage). So, the starch indeed did not have the shape of an uneven exsiccation layer or of a trace contamination. However, the use of a “warp sizing paste” of starch during weaving is not specific for the first century, for it is still used today (products 2-1 and 2-2).
SAPONINS
Saponins from Saponaria (soap-weed) is more specific for ancient technology. For some reason, Rogers didn’t search for saponins (the washing molecules) but only for certain hydrolysis products of Saponaria saponins: reducing pentose sugars. He didn’t find them on non-image-non-scorch fibers (A Chemist’s Perspective, p. 40). Schwalbe&Rogers reported that saponins were not found by PMS (Physical and Chemical, p. 14), but PMS was not really done on controls – the only control was a Raes sample (PMS-paper, p. 2) which released early furfural, the Raes corner being a scorch area ( Internal Selvedge, 2.5.1.). On image or scorch fibers the saponins could or would have been broken down by image formation or scorching. So, in fact, we don’t know whether there are saponins on the background of the Shroud.
SEAM AND STITCHING
Technique
“Dr. Flury-Lemberg found the cloth’s finishing, at its hems, and in the joining seam to have been done using an unusual type of stitching very nearly invisible on one side, and as such closely resembling that of ancient Jewish textiles as found at Masada, the Jewish palace-fortress that was overthrown by the Romans in AD 73, never to be occupied again” (BSTS-newsletter 51). In the Proceedings of the 2000 Turin symposium, Flury-Lemberg shows a drawing of the type of seam (visible in Soons’ presentation, slide 11), and calls it ‘seam type Masada’: “Abb. 3 a Zeichnung: Nahttyp Masada” (Proceedings, p. 34). It might be described as a kind of rolled seam, secured with two lines of tiny overcast stitches. It is interesting that the ancient Egyptians specifically used two lines of overcast stitches to sew on a fringeless braid along an edge of a garment:
“Only a limited range of structural details, such as seams and hems, were used in ancient Egypt. The most common of these were: simple hems, rolled and whipped hems, simple (open) seams, and lap-over seams. Other seams known from the Dynastic period include a form of run-and-fell seam and overcast seams (see Fig. 11.11), but these were rarely used on items of clothing. When a braid was added to a garment, one of several techniques was used, depending on the nature of the braid and the place where it was to be attached. If it was a fringed braid placed at the lower edge of a garment, it would normally be secured with one line of overcast stitching (e.g. Carter no. 367i; Cairo JE 62625). On the other hand, two lines of overcast stitching were used to sew on fringeless braids, whether along an edge of a garment or down the middle” (Ancient Egypt materials and technology, p. 282-283).
The type of seam for the Egyptian fringeless braids is not described or depicted, nor is the Masada-type seam among the depicted seams (Fig. 11.11, p. 283). The Egyptian mending stitches – whipped stitch and running stitch (p. 283) – are different from the non-whipped overcast stitches of the TS seam. Note that the cloth of the Egyptian braids were woven differently from the garment they were sewn on to, as they were meant to be clearly visible decorative elements ( p. 276, figure 11.6).
Moment and reason of seam application
X-ray photography showed “the absence of any evidence of any frayed thread ends along either side of the seam image” and a “detailed thread matching” across the seam (Adler&Whanger, Concerning the Side Strip, with X-ray photos). Also Miller&Pellicori reported the near perfect match in UV, backlight and X-ray (Ultraviolet Photography, p. 85). Flury said that the seam was applied before the lateral hems were, and both were stitched by the same professional hand probably in a single operation (Proceedings 2000, p. 24). Also, the stichting was done from the backside of the cloth (the weft side, where also the hems are), in such way that on the front side the stitches are hardly visible and the seam is invisibly flat (p. 24-25). Moreover, the seam was already planned before weaving, for it is in an internally woven-in selvedge of warp threads that have spun-in cotton (Internal Selvedge, 2.1). The seam thus would have been made at manufacture. It wouldn’t have been a mending, or a rejoining of the two outer parts of the cloth from which an inner third strip had been removed, or even the adding of a decorative braid: it was merely done to have been done. This deliberate extraordinary procedure fits with the strict observance of the religious law of Nu 15,38, which in the Bible that was generally used in first-century Judea literally read that a margin (‘kraspeda’) had to be added to every robe (Nu 15,38 LXX). The first-century Gospel of Matthew says it was custom for Pharisees to “enlarge the borders (‘kraspeda’) of their garments” (Mt 23,5 KJV and WH).
ONE TIME DYE
As a yellow plant dye was found in the starch/starch gum coating (Scientific Method, p. 17-20), and Madder lake particles on the main Shroud (Internal Selvedge, 2.3 and 2.4), it is probable that the Shroud was starched, washed, and then dyed as cloth, without completely removing the starch first. The starch would protect the cloth from wear and tear, but it would not allow warm laundering the dirty cloth without removing the starch and dye together at once. Bleaching or laundering was not allowed for Jewish temple garments – they would simply be cut into wicks for the temple’s oil lamps (Mishneh Torah, Kli Hamikdasch 8, Halacha 4-5) – and the temple was destroyed in 70 AD.
PETALON
3D displays of the TS show a flat, more or less oval object beneath the chin, with protuberant marks that can be read as the Hebrew characters ayin-aleph-nun, almost similar to l X Y in Times New Roman characters (cf. the 1978 photo 210×250 pixels and its 3D display with isolines showing the flatness and the three protuberent characters). The object may be a ‘petalon’ (Ex 28,36 LXX): the distinctive petal-shaped golden ornament of the Jewish high priest (see third-century AD mural) and probably all members of the ruling Jewish Council, the Sanhedrin, consisting of 71 members. The characters l X Y may be the Hebrew numerals 7 and 1 and letter N, an abbreviation of “71 Nesiim” = 71 rulers. First-century historian Josephus said the petalon still existed in the first century (Jewish Antiquities 3,7,6 and 8,3,8). The Jerusalem Gemara (a 350-400 AD commentary on the Mishnah, a 220 AD written version of oral traditions dating from Pharisaic times = 536 BCE – 70 AD) said it was two fingers broad and reached from ear to ear and had protuberant letters (book Kli Hamikdash 9,1-2). Epiphanius said it was worn by James the Just, brother of Jesus (Panarion 29,4,4, p. 125), and Polycrates, stressing the Jewish origin of the Christian Passover date by referring to the evangelist John’s Jewish authority, said in 189-199 AD that the petalon was worn by the priest John the evangelist (Eusebius, Church History 3,31,3), apparently with a different inscription than the two words reserved for the liturgical high priest. The petalon with three letters would be exclusively first-century (6-70 AD), as before 6 AD the petalon was still only worn by the king and the high priest (Epiphanius, Panarion 29,3,3-6 p.124) and after 6 AD the Jews didn’t have a king anymore and the high priest’s liturgical petalon was kept locked up by the Romans (Jew. Antiq. 15,11,4 ad 18,4,3). After 70 AD Jerusalem was devastated by the Romans and there wasn’t a temple or Sanhedrin that ruled Judea anymore.
A petalon on the Shroud would strongly associate it with Jesus, for no other crucifixion victim is known to have been buried in the presence of one, let alone two or three members of the Sanhedrin: Joseph of Arimathea, Nicodemus and John the evangelist. Only John – not Peter – believed Jesus had risen because he saw the shroud (his own temple mantle) rolled up and placed at the head end of the stone bench of the tomb, just as a priest’s temple mantle was always rolled up and placed at the head end of the stone bench in the dormitory of the temple (see my articles The seam and missing corners of the Turin Shroud as characteristics of John Mark’s temple mantle and John Mark – Author of the Gospel of John with Jesus’ mother).
I forgot to stress that the seam rejoins two sections cut from the same cloth (Flury, Proceedings Turin 2000, p. 24 and fig. 3 a, drawing and photo of open TS seam, of which the drawing is also in Soons’ presentation, slide 11). This, and the transverse hems being rolled and stitched across the longitudinal seam (A detailed critical, p. 3 fig. 15), renders the option that the seam was made to hold a supporting rod for display purposes improbable. Thank you, Colin, for reminding me of it.
Actually, the letter ayin ( Y ) means 70 and ayin-aleph ( X Y ) means 71. Hebrew is written from right to left. The nun mark ( l ) on the Shroud is not a final-nun (completely straight: | ), so it isn’t the end of a three-letter word, but rather an abbreviation.
Yes, Adrie, but pardon me asking: how do you know that your letters l X Y have any real existence outside of arbitrary settings on ImageJ 3D software? Who’s to say that the software settings were not played around with until something recognizable as letters (to your modern eye) and/or abbreviation symbols pop up somewhere – “Eureka – a “signal” at last, da da” never mind all the surrounding “noise”.
I’ve previously described a self-disciplined way of using ImageJ to reduce the risk of seeing artefacts. It consists of producing a scorch on linen using a 3D object (e.g. a horse brass), then adjusting ImageJ settings to get as faithful a match as possible to the template, and then applying those same ‘normalised’ settings to the Shroud – or any other image that you believe or hope has embedded or encoded 3D information.
Unless you can produce evidence that your ImageJ settings were normalised in some manner against the “real world” , then I’m afraid I am totally unconvinced that you have the letters l XY. I could play around with the settings and maybe find other letters or marks.
You’re probably going to launch into a long lecture now, but I have to advise you Adrie that despite all your formidable scholarship and impressive attention to detail, from where I am standing you appear sadly to have taken a wrong turning – from the highway of science into an unlit cul-de-sac called agenda-driven obscurantism…
Yes, Colin, I played the settings in Image J, to find the best view for the l X Y . But also the plain STERA photo clearly shows the marks in white, which simply corresponds to the highest level in 3D relative to the surrounding noise. I think it is improbable that the marks themselves are noise.
The Shroud’s fabric has ca. 40 warp threads and 25 weft threads per square cm (Rinaldi, Autogol a Tucson). Each thread (ca. 0.25 mm diameter) consists of 70-120 fibers of 10-20 micrometer diameter (Evidences list, introduction p. 2, and evidence A6), spun in a Z-twist, as opposed to the ordinary Egyptian S-twist (Tyrer, Looking at the Turin Shroud as Textile, p. 20).
The white linen of the temple priest’s garments had to be “bysso retorta” (Ex 39,2.8.22.28 Vulgate), in which “bysso” means the finest/whitest linen or cotton, and “retorta” means ‘twisted backwards’, viz. in a Z-twist, either for a 2-ply twined thread or a 1-ply single thread (The Seam and Missing Corners, p. 16 note 36). The ancient Egyptians were capable of making incredibly fine linen garments, e.g. the girdle of Ramses III has 107-134 warp threads per cm.
Textial expert Vial noted during the 1988 sampling of the Shroud, that “faults in the preparation of the shafts point to a specifically ancient twill weave manufacturing method” and said that “the only European 3.1 chevron twill in linen that stands some comparison
to the Shroud is the canvas of a late 16th. century ‘Last Supper’ painting attributed to Martin
de Vos – and even so its weave is much simpler than that of the Shroud. In effect, he
concludes, the Shroud weave is ‘incomparable’” (BSTS newsletter 26,9, p. 2). Also a sample of a herringbone weave of Z-twisted threads from ca. the second half of the fourteenth century (Victoria & Albert museum), has only 22 warps and 12 wefts per square cm and coarser yarns than those of the Shroud, and thus “is not a candidate for a parallel to the fabric of the Turin Shroud” Tyrer, The textile said to be similar in weave to the Turin Shroud .
WordPress is playing tricks on me: the link to “Tyrer, Looking at the Turin Shroud as Textile” must be http://www.sindone.info/TYRER1.PDF , including the http:// . Otherwise it links to a non-existing bookmark in the main posting.
A post has just appeared on The Other Site that has my name in the title as a “dedication”. It’s from Yannick Clément, still composing and re-composing his Compleat Hagiography of St.Raymond (N.Rogers). If anyone wonders why I choose to stay mute, or nearly so, this passage from the first of his two pdf documents listed should provide a clue:
“Rogers knew perfectly well the chemical structure of the primary cell wall of the linen fiber, even if he didn’t use the term in his writings and nevertheless, he never considered this kind of chemical structure as a valid option to explain the color of the body image on the Shroud. In other words, he never considered the primary cell wall as being the real chromophore of the body image on the Shroud, even though he knew perfectly well the existence of that external part of a linen fiber.”
Holy sh*t…
TH chooses the crudest “bas relief” template imaginable – a metal plate with a shallow, steep-sided recess to represent “relief”. He gets some scorches that not surprisingly have excessive differences in scorch intensity and contrast between the two flat-iron like planes, and then proceeds to enunciate general principles for why the Shroud cannot possibly be a scorch.
Why waste words arguing with someone who is intent only on doing a quick demolition job, taking inexcusable shortcuts experimentally, and liberties with his conclusions? He cannot even be bothered to respond to my 3 postings on his prominently-advertised pdf (The TURIN SHROUD BODY IMAGE: THE SCORCH HYPOTHESIS REVISITED) – see top-right hand corner on Dan Porter’s Home Page .
Here is a link to a a post I did way back in March 2012 , showing a stepwise progression from 1) a more realistic and detailed brass bas-relief template 2) a faint scorch on linen derived therefrom 3) the processing of that scorch in ImageJ software to get a facsimile image that bears a close resemblance to the original – even the lettering is legible. Where’s the excessive contrast? Where are the burned bits?
Now look for the inherent bias. Is it in my experimental design and conclusions or in TH’s? Yes, inherent bias – the hallmark of the pseudo- (agenda-driven) scientist – as if Shroudology has not had enough of those already.
PS: Here’s the kind of facsimile image that is possible if one starts with a proper bas relief template, and uses a faint scorch on one’s linen. One can even read the lettering (“GEORGE VI CORONATION, he of Colin Firth’s “The Ki-Ki-Ki-Ki-ng’s Speech”).
CB wrote: “TH chooses the crudest “bas relief” template imaginable – a metal plate with a shallow, steep-sided recess to represent “relief”. He gets some scorches that not surprisingly have excessive differences in scorch intensity and contrast between the two flat-iron like planes, and then proceeds to enunciate general principles for why the Shroud cannot possibly be a scorch”.
Apparently, CB did not read my paper. Otherwise he would have seen than the major part of it is dealing with the microscopical characteristics of ANY KIND of scorch at fabric and thread level and not with the problem of contrast or relief.
Because my model has no slightly rounded relief, CB thinks that all my observations and photographs are not reliable.
Does CB think that with a slightly rounded relief rather than with a flat template, the microscopical characteristics of the color distribution (and all my observations) at fabric, thread and fiber level should be very different ?
In order to produce an imprint one have to apply a hot surface on the linen.
I did it with the kind of “crudest bas-relief” I used because it was the only mean to test different temperatures, different pressures and different contact times.
Using a “better” bas-relief (in the sense of CB), one can certainly obtain a better “image” but its characteristics at fabric, thread and fiber level cannot be different than the characteristics showed in my paper because the fundamental mechanisms are similar.
And in any case, these characteristics are very different from those observed on the Shroud.
Looking carefully at the slightest King George VI horse brass picture (on the right), I can predict what follows:
With a microscope:
1) In the darkest parts (nose, hear, crown etc.) one will find all the characteristics of the “scorch” as described in my paper;
2) in the lightly scorched areas, one will find all the characteristics of the “light scorch” as described in my paper
3) In the white areas, one will find..no color (contrary to the shroud) or perhaps some fibers described as “very light scorch” in my paper.
To summarize, with a “good” template, one will find all the characteristics I found with my “bad template” but on a single image.
I challenge CB to prove that I am wrong but now with photographs at fabric and thread level.
We will see who is agenda-driven.
TH alias Thibault.
Hooray, a response at last to points made back in October/November. Better late than never.
I propose to deal with them in a series of bite-size instalments. Here’s the first:
Thibault’s pdf: Page 3/24
“The goal was to try to answer to the following question: How could a forger get the shading necessary to obtain an image?”
(Note: he says “shading”, not microscopical characteristics)
“It is clear that the parameters are: the temperature of the hot bas-relief, the contact time and the contact “intensity”.
Yes, the contact intensity, which depends among other things on the angle of contact. There are only two angles of contact with Thibault’s template – 0 degrees (the two facing planes) and 90 degrees (the shallow ledge between the two planes). There is nothing between those two extremes to give the subtle bas relief effect from what a physicist might describe as resolved force/pressure vectors – one that I was as such pains to describe in my reply, using home-made graphics in MS Paint to ensure that my point was not lost.
Image from part 1 (October 23) of last year’s 3 part response to Thibault.
Clearly my point was lost, in spite of all my best efforts.
“What is interesting here is that this can be obtained naturally by using a bas-relief.”
Yes!
“If the forger used a uniformly heated bas-relief and applied a linen on it, it is possible to have a
contact everywhere (necessary condition). Naturally, the contact in the flat areas or small
depressions of the bas-relief would be light while the contact with higher areas (like the nose)
would be more intense.”
Yes, but it’s not only height differences above a reference plane that one has to consider with a bas relief. It’s the angle that each point on the template makes with the linen, which can take any value between 0 and 90 degrees. 0 degrees means “square on”, maximum pressure. 90 degrees means tangential to the linen, in contact perhaps, merely brushing so to speak, so virtually no application pressure. That’s a major blind spot being revealed here.
“This last experiment shows how difficult it is to manage the temperature to obtain something
resembling the TS image shading. On the image on the right, the lateral bands have the right yellow…”
We’re only on page 5, and here we see the author attempting to demolish the validity of the scorch model. He does not have a proper bas relief with rounded contours presenting a full range of contact angles to produce a subtle image with gradations of intensity and contrast, yet here he is making invidious comparisons with the TS images which he has not even presented at this stage (they appear much later in the pdf, ie. the Mark Evans micrographs on page 15-17).
Page 6: “This problem of contrast has already been discovered by Jackson….”
He is now looking to another author to support his unsupported case, based on an inappropriate template. This is the kind of thing one would expect to find in Discussion, not inserted into Results, Page 6 of a 24 page paper no less. This is clear impatience on Thibault’s part to start his summing up and dismissal of scorching. This is why I say his paper is agenda-driven. Scientists evaluate all the evidence in their results before arriving at a conclusion: they don’t go proselytising or even hinting at conclusions until the … Conclusions section. Premature conclusions have no place in a Results section. They place a huge question mark over the author’s objectivity.
“This high intensity of shading at contact points was necessary in order for the more distant parts of the bas-relief to record their patterns on the cloth”
With a proper dome-shaped bas relief, it is not necessary to rely on heat conduction for the less superficial parts of the template to make contact. The cloth drapes around the contours allowing contact in most places. The only places not make contact would be narrow and/or deep grooves (“crevices”) where the cloth bridges the gap. But there are plenty of examples on the TS where there appears to be bridging, and I don’t just mean the gaps between fingers but that sizeable non-image-bearing area of abdomen around the crossed hands. Note that I am referring to macroscopic aspects that everyone is familiar with. Microscopic characteristics can wait till later.
Reminder from earlier (Page 6): “This problem of contrast has already been discovered by Jackson”
There is NO self-evident problem of contrast. Any problem, if it exists, is likely to be due to use of a crude template without a dome shape and gentle rounded contours (we’ll address microscopic aspects later).
So far I have addressed the issue raised at the start, namely whether a forger could get the shading necessary for the image. One should address this issue first, and separately, before microscopical details. Repeat: we are discussing the macroscopic shading and contrast. There is no need to introduce microscopical criteria at this stage. They can wait.
Thibault needs to repeat his experiments with a proper template, and review his opinions re shading and contrast. He should keep his microscope in the cupboard for now – one thing at a time.
I’ll add another instalment tomorrow, or Monday at the latest. Promise.
Pingback: Thibault Responds to Colin « Shroud of Turin Blog
Part 2 of my reply to Thibault.
“Apparently, CB did not read my paper. Otherwise he would have seen than the major part of it is dealing with the microscopical characteristics of ANY KIND of scorch at fabric and thread level and not with the problem of contrast or relief.”
OK, now we have addressed the macroscopic aspects, we can deal with the microscopic. It’s immaterial as to which is the major or minor part of the paper. They are both important, but it’s customary to start with the macroscopic aspects first, especially when we are asked to see things from a forger’s point of view – someone wanting to get subtle gradations of contrast.
Here btw is a “macroscopic” view of a scorch off a horse-brass template (beats a chunk of discarded steel anyday) that has been 3D enhanced in ImageJ. I see no problem with contrast. Do you, does anyone, apart from a blotch on the nose – that could be pixellation noise.
“Because my model has no slightly rounded relief, CB thinks that all my observations and photographs are not reliable.”
Correct. They are not reliable. Why, when others have used bas-relief with recognizable images, with subtleties of shading and contrast, do you decide to use a flat chunk of metal with no image as such, merely a sunken plane. It makes no sense to embark on the microscopy if the macroscopic modelling is wrong. By you own admission you have excessive contrast, so it’s hardly surprising that the microscopy should exaggerate differences between scorched, unscorched and lightly scorched. Intentionally or otherwise, you have arranged matters to get a negative result, namely that a scorch is a bad model of the Shroud. No, a bad scorch is a bad model for the Shroud,
“Does CB think that with a slightly rounded relief rather than with a flat template, the microscopical characteristics of the color distribution (and all my observations) at fabric, thread and fiber level should be very different ?
We are talking about modelling a subtle image, so it’s anyone guess what is meant when you ask if I would expect a difference at the microscopic level between a rounded bas relief and a flat piece of metal. If one image has subtle contrast differences and the other has harsher differences, with excessive scorching, then yes, of course one would expect differences. But at this stage, we are not trying to make predictions, since we have little or no experience of scorching. Where are there published papers? So let’s put our ideas and presuppositions to one side, and simply look at the model scorches in THIS paper, with its methodology, and see how they compare with the Shroud. Are there irreconcilable differences?
“In order to produce an imprint one has to apply a hot surface on the linen.
I did it with the kind of “crudest bas-relief” I used because it was the only mean to test different temperatures, different pressures and different contact times.”
Even if that were the case, i.e. that a proper bas relief could not have been used, that would not justify a model that was unsuited to the task in hand. But I don’t accept anyway the claim that one had to use a flat sheet of metal. What is it you were not able to do with a bas rellief that you could do with your quirky flat iron.? Different temperatures? Simply do serial imprinting as the template cools down, as I have done, to get a range of scorch-intensities. Different pressures? Press the hot template into an underlay with a metal implement applying light or heavy pressure. Different contact times? Have a pair of pliars or similar handy to remove the template from the linen at different times. Grab it edge on it necessary.
“Using a “better” bas-relief (in the sense of CB), one can certainly obtain a better “image” but its characteristics at fabric, thread and fiber level cannot be different than the characteristics showed in my paper because the fundamental mechanisms are similar.”
The fundamental “mechanism” may be similar at the molecular level, but image imprinting is not just about fundamental science, anymore than any other kind of image-making. It’s as much to do with technique. Much emphasis was placed earlier on alleged differences in “contrast”. But they can arise without any difference in the mechanism of scorching at the molecular level– merely by differences in application time, pressure, temperature and ANGLE of contact, as determined by the contours of the template (admittedly out of the operative’s control, once the template has been cast, but by the same token making the image technique highly reproducible – imprinting as opposed to painting or other artwork.
And in any case, these characteristics are very different from those observed on the Shroud.
Yes, but we are not comparing like with like. Your image was probably a few hours old before being photographed. The Shroud’s image is centuries old. One has had negligible wear and tear. The other has probably had extensive wear and tear. We know from STURP’s work with sticky tape that the (surviving) image fibres are weaker and more prone to breakage than non-image areas (a major and much-neglected observation). It is hardly surprising that the Shroud would have lost much of its image by now if the latter were, as I believe, a scorch, simply via random breakage and detachment of the more scorched, more brittle fibres. That explains why the modern Shroud image is now so indistinct. It was not always that way. It would not have been the crowd-puller if that had been the case (scores of people occasionally died in the crush to see it, even glimpsed from a distance).
“Looking carefully at the slightest King George VI horse brass picture (on the right), I can predict what follows:
With a microscope:
1) In the darkest parts (nose, hear, crown etc.) one will find all the characteristics of the “scorch” as described in my paper;
2) in the lightly scorched areas, one will find all the characteristics of the “light scorch” as described in my paper
3) In the white areas, one will find..no color (contrary to the shroud) or perhaps some fibers described as “very light scorch” in my paper.”
Yes, my image is a model scorch, and so is yours, so there are no grounds for thinking there will be a major qualitative differences between yours and mine at the microscopic level. I have never questioned the veracity of your images of the scorches under the microscope. Why should I? Mine differ little from yours. Why should they? A scorch is a scorch. The key question is how BOTH our scorch images compare with the Shroud.
“To summarize, with a “good” template, one will find all the characteristics I found with my “bad template” but on a single image.
Meaning what precisely?
“I challenge CB to prove that I am wrong but now with photographs at fabric and thread level.”
You have ducked (here) out of the major question – why you have rejected scorching, based on model scorches v the Shroud.
I can easily summmarise my position: at the gross (macroscopic) level the Shroud image has many of the characteristics of a scorch, as STURP members noted. At the microscopic level there may be differences, but whether they are the result of the image being something other than a scorch or whether they represent the effect of ageing and degradation is a moot point. You have done nothing to throw light on that question. What one cannot do is dismiss scorching without considering ageing differences. (It is possible that with time (no pun intended) ageing differences can also modelled after a fashion, e.g. with accelerated ageing, induced by chemical agents, and/or accelerated mechanical wear and tear.
I shall write a third instalment, commenting on the organization of Thibault’s pdf, with its partisan comment not just in the Conclusions, but from the very start, in the Introduction no less, and even intruding into so-called Results. It’s not a top priority for me – having published a detailed rebuttal of Thibaults paper back in October/November and having been totally ignored all these weeks. It’s now Thibault’s turn to wait – I have other interests to pursue (see current posting on that alleged Anglo-Saxon conquest) and to be honest am not hugely enamoured of Thibault’s kind of “science”. It lacks objectivity. Science without objectivity tends to morph imperceptibly into pseudo-science, as is painfully apparent to at least one retired scientist (me), reading that pdf of his.
Colin,
You are right in writing: “The key question is how BOTH our scorch images compare with the Shroud.”
The only problem is that you never showed your scorch image at fabric and thread level.
This is the key.
You can compare my scorch image with the TS at fabric and thread level.
I can not compare your own scorch image with the TS at fabric and thread level.
You published your pictures of scorched linen back in October/November, Thibault, before I had acquired my own microscope. I had no problem whatsoever with them then, a position that has not changed since doing my own microscopy. That is why I have not challenged them, which is why I have not felt in the least bit obliged or even motivated to produce my own. Why are you so keen for me to publish my own pictures when they are essentially the same as yours?
Duplicating other people’s work is not something that appeals to me greatly, unless I suspect it to be misreported. My interest has been with the reported fragility of Shroud image fibres which I consider consistent with thermal degradation conferring brittleness. My interest is in understanding where the weak points exist in a scorched fibre. So yes, I HAVE published pictures of scorched linen fibres, suggesting initially that the dislocations were the weak point, followed later by a suggestion that the 15% non-cellulosic polysaccharides of the secondary cell wall are additional sites of thermal degradation within the core of the fibre, separate from the more superficial image-capture zone.
As I am shortly to do an exposé of a best-seller that fails to document the author’s chief claim, while padding out the rest of the volume with a (re) analysis of other people’s results, I am hardly likely to do the same now, am I.?
I repeat: I have no problem whatsoever with your scorch photomicrographs. The difference is that I believe they can be reconciled with the Mark Evans photomicrographs, once age-related degradation and mechanical wear and tear are taken into account.
Scientific research is about unremitting focus on the essentials. You will not succeed in diverting me to matters that I regard as non-essential, least of all sterile duplication of findings I do not dispute. It’s the correct interpretation of those findings that is the important thing…
This retired science bod is frankly unenamoured of the partisan way in which you have interpreted your findings – using them to bolster yours (and others) anti-scorch position, no doubt because it fits a 14th century rather than your preferred 1st century provenance, with the imprinting of an image from a heated template as distinct from a real human being. (That would not preclude ‘death mask’ technology for casting the effigy-cum-template from a real human being, whether alive or dead).
Good news.
You now seem to agree that you saw what I saw but on your own “good” template.
Therefore I have to conclude that finally my template was not so bad as a model template.
OK.
Now the problem is the interpretation.
Let’s go step by step, forgetting for the moment the (true) question of the fragility of the Shroud fibers.
You wrote: “I repeat: I have no problem whatsoever with your scorch photomicrographs. The difference is that I believe they can be reconciled with the Mark Evans photomicrographs, once age-related degradation and mechanical wear and tear are taken into account.”
I believe that there is no mean for any kind of age-related degradation and mechanical wear and tear to transform a scorch area into a TS image area as seen on ME photomicrographs for many reasons.
Before explaining why (although the reasons can be easily found in my paper), I would like to see a demonstration of your hypothesis.
Oh come now, Thibault. Even allowing for the fact that English is not your first language, you must know that one does not “demonstrate” a hypothesis, least of all a working hypothesis.
One may patiently assemble evidence that supports or contradicts it, but researching a hypothesis can be a life’s work, and even then it may only be elevated to theory, not established fact.
Neither do I care for the line you are now taking on my acceptance of your photographs. No, I do not think we have established common ground at all. I work with faint scorches, with a view to modelling the Shroud. You produce scorches with excessive contrast, which while having regions of faint scorching – like mine – also have regions of excessive scorching, which you claimed must ALWAYS be a feature of scorching and ipso facto rules out scorching as the mechanism by which the Shroud image was produced.
I merely said I accepted your photographs (believing them to be consistent with your poor choice of template). I did not say that I accepted them as a valid model for the Shroud, which is why I regard your microscopy as being of limited validity re modelling, and indeed a potential source of misunderstanding, especially in view of the agenda-driven way you organized and presented your findings. So please don’t conflate your experimental approach and conclusions with my own. My interest is in modelling the Shroud, not in seeking to demolish the scorch hypothesis on the back of one or two hasty, ill-designed experiments.
PS: to Thibault:
Of course, it’s for you to decide how to write your own papers (and for me to decide how to arrive at a personal view).
My personal view would have been more favourable if you had not introduced your paper with the fluorescence problem, as if to suggest that the weight of evidence was already against scorching.
There are lots of unknowns re fluorescence, notably the precise chemical nature of the fluorescent pyrolysis products at the molecular level, their chemical stability, their volatility. The TS image might possibly have fluoresced under uv when first formed, but if that were due to furfural, say, which you cite, then it’s unlikely there would be any still around after centuries of storage, especially after exposure to high temperature (1532 fire).
Furfural is a small molecule (C5H4O2) with a relatively low boiling point (162 degrees C) and thus an appreciable vapour pressure at normal temperatures. More importantly, you made no attempt to monitor fluorescence in your own paper, so had nothing new to offer. Why then introduce fluorescence into your Introduction, except to signal that there was in your view a corpse called the scorch hypothesis, one needing a decent burial, and you were about to bang the last nails into the coffin?
Sorry, I have no time for your brand of agenda-driven pseudo-science Thibault. Stick to your chosen profession: diagnosing and treating your patients’ ailments. You are under a serious misapprehension regarding the scientific method. My advice to you would be to leave science to the scientists.
PPS Observant readers may note I have manicured the site’s appearance today: there is a new less aggressive title and accompanying message, new background to fill in blank space to left and right of the content. The repeating images in that previously blank space were obtained by taking a ShroudScope image of the face, adding some contrast, then showing the result of light/dark reversal (“inversion”) in ImageJ, followed by 3D enhancement. So folk can see at a glance the iconic features of the Shroud image that arise through modern stepwise processing (conversion of the “photographic negative”, correction, thermographic negative, to a positive, as per Secondo Pia, then the demonstration of so-called “encoded 3D information”. Forget the mystique: in fact nothing is encoded – certainly not digitally – except by via analogue/tactile reproduction, like a gramophone record in a groove. The ImageJ software simply reads image intensity as height on the z axis above the xy plane). More importantly, one can see that the same transformations are attainable in principle by starting with a thermal imprint (“scorch”) from a heated template (see banner, with my Ghanaian trinket as bas-relief template).
I ask everybody to read my previous message in which I ask CB to show or at least to provide some explanations and demonstrations regarding his hypothesis: ” I have no problem whatsoever with your scorch photomicrographs. The difference is that I believe they can be reconciled with the Mark Evans photomicrographs, once age-related degradation and mechanical wear and tear are taken into account”.
Look at CB answer: a contemptuous reply that is not a reply but an infamous meaningless mixture that has nothing to do with my proposal.
No demonstration, no explanation, no experiment, no proposal….
Now who is agenda-driven ? Who follows the true scientific method ?
CB who wants AT ANY PRICE to save his scorch hypothesis (using more and more ad-hoc incredible allegations (snow, …) and does not even answer to my polite questions/proposals above, or myself ?
Sorry, I have no time for CB brand of agenda-driven pseudo-science. CB is under a serious misapprehension regarding the scientific method.
At least I have a hypothesis, one that can account for the colour, the reflectance absorption spectrum, the light/dark reversed image, the responsiveness to 3D enhancement, the preferential location on the crowns of the weave, the known chemistry of polysaccharide pyrolysis etc etc. (Need I continue?) For his part, what does Thibault have? Nothing – no hypothesis to account for a sheet of linen with an unusual sepia-coloured image – but a material object nevertheless. All he has to offer is a pseudo-scientific pdf attacking a plausible and promising hypothesis defended with known physics and chemistry, simply because it doesn’t fit with his preconceptions of authenticity.
The Thibaults of this world are not part of the solution. They are part of the problem. That is why I set up this site, a spin-off from sciencebuzz.com, to root out the pseudo-science of Shroudology – and for no other reason. Pseudo-science makes a mockery of the scientific method, the latter depending on people willing to go beyond the known facts and advance a hypothesis. Repeat: a hypothesis cannot be demonstrated. To suggest otherwise betrays a profound ignorance of the scientific method. A hypothesis is a construct, a working model, one that serves to stimulate new thinking, new experimental approaches. This blog has over the weeks and months proposed any number of suggestions for new experimental approaches, based on the unproven supposition (i.e. hypothesis) that the TS image is a thermal imprint.
That is all I have to say to Thibault for now. He can rest assured that I will not hesitate to speak again should he produce any more pdf reports that lack the essential objectivity of science, ones that simply attempt to dismiss an existing hypothesis with phoney argument, creating difficulties, nay, insuperable problems, that are entirely in the mind of the author.
Repeat: there is NO insuperable problem regarding scorch contrast or intensity, as the photograph below demonstrates. The faintest scorches, close to the limit of visibility, give very satisfactory light/dark reversed and 3D-enhanced images. It was galling in the extreme for Thibault to propose otherwise, based on a totally unsuitable choice of template, and failing to mention anywhere the scores of postings I have done using more carefully-chosen bas relief templates. But then they undermine his simplistic and absurdly dogmatic thesis, don’t they?
Left: scorch imprints with decreasing intensity, obtained by serial imprinting from heated horse brasses as they cool down.
Right: the same photograph, after negative->positive conversion and semi-3D enhancement in ImageJ software.
Scorches of fainter or intermediate intensity give the best 3D facsimiiles – there is no problem due to one part of the scorch image being too intense relative to another. The “contrast problem” is a total red herring – one that needs to be quickly retracted by its sponsor(s). It only exists due to unsuitable choice of template or the manner in which it is deployed.